By agreement with the publisher, this book is accessible by the search feature, but cannot be browsed.
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Cooper GM. The Cell: A Molecular Approach. 2nd edition. Sunderland (MA): Sinauer Associates; 2000.

The Cell: A Molecular Approach. 2nd edition.
Show detailsThe carrier and channel proteins discussed in the preceding section transport small molecules through the phospholipid bilayer. Eukaryotic cells are also able to take up macromolecules and particles from the surrounding medium by a distinct process called endocytosis. In endocytosis, the material to be internalized is surrounded by an area of plasma membrane, which then buds off inside the cell to form a vesicle containing the ingested material. The term “endocytosis” was coined by Christian deDuve in 1963 to include both the ingestion of large particles (such as bacteria) and the uptake of fluids or macromolecules in small vesicles. The former of these activities is known as phagocytosis (cell eating) and the latter as pinocytosis (cell drinking).
Phagocytosis
During phagocytosis, cells engulf large particles such as bacteria, cell debris, or even intact cells (Figure 12.34). Binding of the particle to receptors on the surface of the phagocytic cell triggers the extension of pseudopodia—an actin-based movement of the cell surface, discussed in Chapter 11. The pseudopodia eventually surround the particle and their membranes fuse to form a large intracellular vesicle (>0.25 μm in diameter) called a phagosome. The phagosomes then fuse with lysosomes, producing phagolysosomes in which the ingested material is digested by the action of lysosomal acid hydrolases (see Chapter 9). During maturation of the phagolysosome, some of the internalized membrane proteins are recycled to the plasma membrane, as discussed in the next section for receptor-mediated endocytosis.
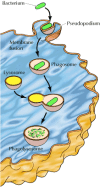
Figure 12.34
Phagocytosis. Binding of a bacterium to the cell surface stimulates the extension of a pseudopodium, which eventually engulfs the bacterium. Fusion of the pseudopodium membranes then results in formation of a large intracellular vesicle (a phagosome). (more...)
The ingestion of large particles by phagocytosis plays distinct roles in different kinds of cells (Figure 12.35). Many amoebas use phagocytosis to capture food particles, such as bacteria or other protozoans. In multicellular animals, the major roles of phagocytosis are to provide a defense against invading microorganisms and to eliminate aged or damaged cells from the body. In mammals, phagocytosis is the function of primarily two types of white blood cells, macrophages and neutrophils, which are frequently referred to as “professional phagocytes.” Both macrophages and neutrophils play critical roles in the body's defense systems by eliminating microorganisms from infected tissues. In addition, macrophages eliminate aged or dead cells from tissues throughout the body. A striking example of the scope of this activity is provided by the macrophages of the human spleen and liver, which are responsible for the disposal of more than 1011 aged blood cells on a daily basis.

Figure 12.35
Examples of phagocytic cells. (A) An amoeba engulfing another protist. (B) Macrophages ingesting red blood cells. False color has been added to the micrograph. (A, R. N. Band and H. S. Pankratz/Biological Photo Service; B, courtesy of Joel Swanson.)
Receptor-Mediated Endocytosis
In contrast to phagocytosis, which plays only specialized roles, pinocytosis is common among eukaryotic cells. The best-characterized form of this process is receptor-mediated endocytosis, which provides a mechanism for the selective uptake of specific macromolecules (Figure 12.36). The macromolecules to be internalized first bind to specific cell surface receptors. These receptors are concentrated in specialized regions of the plasma membrane, called clathrin-coated pits. These pits bud from the membrane to form small clathrin-coated vesicles containing the receptors and their bound macromolecules (ligands). The clathrin-coated vesicles then fuse with early endosomes, in which their contents are sorted for transport to lysosomes or recycling to the plasma membrane.

Figure 12.36
Clathrin-coated vesicle formation. (A) Extracellular macromolecules (ligands) bind to cell surface receptors that are concentrated in clathrin-coated pits. These pits bud from the plasma membrane to form intracellular clathrin-coated vesicles. (B) Electron (more...)
The uptake of cholesterol by mammalian cells has provided a key model for understanding receptor-mediated endocytosis at the molecular level. Cholesterol is transported through the bloodstream in the form of lipoprotein particles, the most common of which is called low-density lipoprotein, or LDL (Figure 12.37). Studies in the laboratories of Michael Brown and Joseph Goldstein demonstrated that the uptake of LDL by mammalian cells requires the binding of LDL to a specific cell surface receptor that is concentrated in clathrin-coated pits and internalized by endocytosis. As discussed in the next section, the receptor is then recycled to the plasma membrane while LDL is transported to lysosomes, where cholesterol is released for use by the cell.

Figure 12.37
Structure of LDL. Each particle of LDL contains approximately 1500 molecules of cholesteryl esters in an oily core. The core is surrounded by a coat containing 500 molecules of cholesterol, 800 molecules of phospholipid, and one molecule of apoprotein (more...)
The key insights into this process came from studies of patients with the inherited disease known as familial hypercholesterolemia. Patients with this disease have very high levels of serum cholesterol and suffer heart attacks early in life. Brown and Goldstein found that cells of these patients are unable to internalize LDL from extracellular fluids, resulting in the accumulation of high levels of cholesterol in the circulation. Further experiments demonstrated that cells of normal individuals possess a receptor for LDL, which is concentrated in coated pits, and that familial hypercholesterolemia results from inherited mutations in the LDL receptor. These mutations are of two types. Cells from most patients with familial hypercholesterolemia simply failed to bind LDL, demonstrating that a specific cell surface receptor was required for LDL uptake. In addition, a few patients were identified whose cells bound LDL but were unable to internalize it. The LDL receptors of these patients failed to concentrate in coated pits, providing direct evidence for the central role of coated pits in receptor-mediated endocytosis.
The mutations that prevent the LDL receptor from concentrating in coated pits lie within the cytoplasmic tail of the receptor and can be as subtle as the change of a single tyrosine to cysteine (Figure 12.38). Further studies have defined the internalization signal of the LDL receptor as a sequence of six amino acids, including the essential tyrosine. Similar internalization signals, frequently including tyrosine residues, are found in the cytoplasmic tails of other receptors taken up via clathrin-coated pits. These internalization signals bind to adaptor proteins, which in turn bind clathrin on the cytosolic side of the membrane (Figure 12.39), similar to the way in which clathrin-coated vesicles form during the transport of lysosomal hydrolases from the trans Golgi network (see Figure 9.31). Clathrin assembles into a basketlike structure that distorts the membrane, forming invaginated pits. A GTP-binding protein, called dynamin, assembles into rings around the necks of these invaginated pits, eventually leading to the release of coated vesicles inside the cell.

Figure 12.38
The LDL receptor. The LDL receptor includes 700 extracellular amino acids, a transmembrane α helix of 22 amino acids, and a cytoplasmic tail of 50 amino acids. The N-terminal 292 amino acids constitute the LDL-binding domain. Six amino acids within (more...)
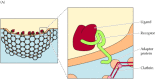
Figure 12.39
Formation of clathrin-coated pits. (A) Adaptor proteins bind both to clathrin and to the internalization signals present in the cytoplasmic tails of receptors. (B) Electron micrograph of a clathrin-coated pit showing the basketlike structure of the clathrin (more...)
Receptor-mediated endocytosis is a major activity of the plasma membranes of eukaryotic cells. More than 20 different receptors have been shown to be selectively internalized by this pathway. Extracellular fluids are also incorporated into coated vesicles as they bud from the plasma membrane, so receptor-mediated endocytosis results in the nonselective uptake of extracellular fluids and their contents (fluid phase endocytosis), in addition to the internalization of specific macromolecules. Coated pits typically occupy 1 to 2% of the surface area of the plasma membrane and are estimated to have a lifetime of 1 to 2 minutes. From these figures, one can calculate that receptor-mediated endocytosis results in the internalization of an area of cell surface equivalent to the entire plasma membrane approximately every 2 hours.
A variety of studies indicate that cells also possess clathrin-independent endocytosis pathways. For example, fluids and some membrane-bound molecules continue to be endocytosed under experimental conditions that inhibit endocytosis from clathrin-coated pits. One pathway of clathrin-independent endocytosis involves the uptake of molecules in small invaginations of the plasma membrane (50 to 80 nm in diameter) called caveolae (Figure 12.40). Caveolae are enriched in lipid rafts of cholesterol and sphingolipids and possess a distinct coat formed by a protein called caveolin. They have been implicated in cell signaling and a variety of transport processes, including endocytosis. In addition, large vesicles (0.15 to 5.0 μm in diameter) can mediate the uptake of fluids in a process known as macropinocytosis. Thus, while clathrin-dependent endocytosis clearly provides a major pathway for the uptake of both fluids and specific macromolecules, cells also use several possible clathrin-independent mechanisms.

Figure 12.40
Caveolae. Electron micrographs of caveolae. (A, courtesy of R. G. W. Anderson/University of Texas Southwestern Medical School-Dallas; B, courtesy of John E. Heuser, Washington University School of Medicine.)
Protein Trafficking in Endocytosis
Following their internalization, clathrin-coated vesicles rapidly shed their coats and fuse with early endosomes, which are vesicles with tubular extensions located at the periphery of the cell. The specificity of fusion of endocytic vesicles with endosomes is determined by interactions between complementary pairs of transmembrane proteins of the vesicle and target membranes (v-SNAREs and t-SNAREs) and by Rab GTP-binding proteins, as discussed in Chapter 9. The early endosomes serve as a sorting compartment, from which molecules taken up by endocytosis are either recycled to the plasma membrane or transported to lysosomes for degradation. In addition, the early endosomes of polarized cells can transfer endocytosed proteins between different domains of the plasma membrane—for example, between the apical and basolateral domains of epithelial cells.
An important feature of early endosomes is that they maintain an acidic internal pH (about 6.0 to 6.2) as the result of the action of a membrane H+ pump. This acidic pH leads to the dissociation of many ligands from their receptors within the early endosome compartment. Following this uncoupling, the receptors and their ligands can be transported to different intracellular destinations. A classic example is provided by LDL, which dissociates from its receptor within early endosomes (Figure 12.41). The receptor is then returned to the plasma membrane via transport vesicles that bud from the tubular extensions of endosomes. In contrast, LDL is transported (along with other soluble contents of the endosome) to lysosomes, where its degradation releases cholesterol.

Figure 12.41
Sorting in early endosomes. LDL bound to its receptor is internalized in clathrin-coated vesicles, which shed their coats and fuse with early endosomes. At the acidic pH of early endosomes, LDL dissociates from its receptor and the endocytosed materials (more...)
Recycling to the plasma membrane is the major fate of membrane proteins taken up by receptor-mediated endocytosis, with many receptors (like the LDL receptor) being returned to the plasma membrane following dissociation of their bound ligands in early endosomes. The recycling of these receptors results in the continuous internalization of their ligands. Each LDL receptor, for example, makes a round-trip from the plasma membrane to endosomes and back approximately every 10 minutes. The importance of the recycling pathway is further emphasized by the magnitude of membrane traffic resulting from endocytosis. As already noted, approximately 50% of the plasma membrane is internalized by receptor-mediated endocytosis every hour and must therefore be replaced at an equivalent rate. Most of this replacement is the result of receptor recycling; only about 5% of the cell surface is newly synthesized per hour.
Ligands and membrane proteins destined for degradation in lysosomes are transported from early endosomes to late endosomes, which are located near the nucleus (see Figure 12.41). Transport from early to late endosomes is mediated by the movement of large endocytic carrier vesicles along microtubules. The late endosomes are more acidic than early endosomes (pH about 5.5 to 6.0) and, as discussed in Chapter 9, are able to fuse with transport vesicles carrying lysosomal hydrolases from the Golgi apparatus. Late endosomes then mature into lysosomes as they acquire a full complement of lysosomal enzymes and become still more acidic (pH about 5). Within lysosomes, the endocytosed materials are degraded by the action of acid hydrolases.
Although many receptors (like the LDL receptor) are recycled to the plasma membrane, others follow different fates. Some are transported to lysosomes and degraded along with their ligands. For example, the cell surface receptors for several growth factors (discussed in the next chapter) are internalized following growth factor binding and eventually degraded in lysosomes. The effect of this process is to remove the receptor-ligand complexes from the plasma membrane, thereby terminating the response of the cell to growth factor stimulation—a phenomenon known as receptor down-regulation.
A specialized kind of recycling from endosomes plays an important role in the transmission of nerve impulses across synapses (Figure 12.42). As discussed earlier in this chapter, the arrival of an action potential at the terminus of most neurons signals the fusion of synaptic vesicles with the plasma membrane, releasing the neurotransmitters that carry the signal to postsynaptic cells. The empty synaptic vesicles are then recovered from the plasma membrane in clathrin-coated vesicles, which fuse with early endosomes. The synaptic vesicles are then regenerated directly by budding from endosomes. They accumulate a new supply of neurotransmitter and recycle to the plasma membrane, ready for the next cycle of synaptic transmission.
Figure 12.42
Recycling of synaptic vesicles.
In polarized cells (e.g., epithelial cells), internalized receptors can also be transferred across the cell to the opposite domain of the plasma membrane—a process called transcytosis. For example, a receptor endocytosed from the basolateral domain of the plasma membrane can be sorted in early endosomes for transport to the apical membrane. In some cells, this is an important mechanism for sorting membrane proteins (Figure 12.43). Rather than being sorted for delivery to the apical or basolateral domains in the trans Golgi network (see Figure 9.28), proteins are initially delivered to the basolateral membrane. Proteins targeted for the apical membrane are then transferred to that site by transcytosis. In addition, transcytosis provides a mechanism for the transfer of extracellular macromolecules across epithelial cell sheets. For example, many kinds of epithelial cells transport antibodies from the blood to a variety of secreted fluids, such as milk. The antibodies bind to receptors on the basolateral surface and are then transcytosed along with their receptors to the apical surface. The receptors are then cleaved, releasing the antibodies into extracellular secretions.

Figure 12.43
Protein sorting by transcytosis. A protein destined for the apical domain of the plasma membrane is first transported from the Golgi apparatus to the basolateral domain. It is then endocytosed and selectively transported to the apical domain from early (more...)
Box
Key Experiment: The LDL Receptor.
- Endocytosis - The CellEndocytosis - The Cell
- Books and Other Individual Titles on the Internet - Citing MedicineBooks and Other Individual Titles on the Internet - Citing Medicine
- UBE2MP1 ubiquitin conjugating enzyme E2 M pseudogene 1 [Homo sapiens]UBE2MP1 ubiquitin conjugating enzyme E2 M pseudogene 1 [Homo sapiens]Gene ID:606551Gene
- H3c2 H3 clustered histone 2 [Mus musculus]H3c2 H3 clustered histone 2 [Mus musculus]Gene ID:319150Gene
- LRRC49 leucine rich repeat containing 49 [Homo sapiens]LRRC49 leucine rich repeat containing 49 [Homo sapiens]Gene ID:54839Gene
Your browsing activity is empty.
Activity recording is turned off.
See more...