NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Madame Curie Bioscience Database [Internet]. Austin (TX): Landes Bioscience; 2000-2013.
In this overview we describe surgical procedures and hypertermic-isolated limb perfusion technique for the treatment of in transit metastases from melanoma and sarcoma of the limbs. We also briefly analyze the rationale of limb perfusion procedure. The procedures are divided, for teaching purpose, in three phases (surgical procedure, perfusion time, reconstructive phase). Finally we present a brief summary of our casuistry and results obtained in the treatment of sarcoma and melanoma. We have performed 91 limb perfusions on 86 patients (5 patients have been treated twice). We obtained an objective response on 93.6% of patients with in-transit metastases from melanoma (45.5% presented a complete response and 48.1% a partial response). About sarcoma of limbs, we reached an objective response on 80% of patients. Side effects have been mild and not life threatening (e.g., edema of the limb, leucopenia and a compartment syndrome).
Introduction and Indications of Limb Perfusion
The principles underlying the synergistic effects of cytostatic drugs and hyperthermia have been extensively described on previous chapters; briefly, we must underline that isolated limb perfusion offers two main pharmacokinetics advantages compared to sistemic neoplastic treatment: (a) high drug concentration in the tumor area, (b) low systemic toxicity. Those important effects have been clinically applied initially for in-transit metastases from melanoma. In this condition, melanoma disseminates through the whole limb from the initial site of melanoma to the regional lymph nodes. This situation is not easy-managing with conventional surgical or chemotherapeutic treatments but is still a locally-advanced disease. Isolated limb perfusion allows to reach good results on survival and quality of patient's life, without the toxic effects of systemic chemotherapy. Limb perfusion consists of three phases:
- Surgical demolishing phase (node dissection and vessels preparation)
- Perfusion phase
- Reconstructive phase
Basically, the treatment consists in isolating the limb from systemic circulation and perform a limb perfusion with cytostatic drug for 60 minutes with extracorporeal circulation technique (ECC) (Fig. 1).
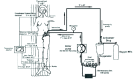
Figure 1
Circuit scheme of isolated limb perfusion.
At the beginning of perfusion phase, the blood of the limb is heated; when tumor site and the whole limb reach an homogeneous temperature of 41.3-41.5°C, the drugs are injected into perfusion circuit at high concentration with low side effects for the rest of the body. The pharmacological benefit is linked not only to high concentration but also to a continuous circulation of drugs in the limb that increases the cytostatic uptake from tumor. As previously described, hyperthermia interacts with anticancer drugs at multiple levels. A direct effect is a selective damage induced on tumor cells due to decreased adaptability of tumor vessels to heat administration. This effect expresses in increased heat-entrapment in tumor areas than in normal tissue, rendering hyperthermia a selective therapy. Another effect is related to increased permeability of tumor-cell membrane, induced by heat, that allows accumulation of drugs inside cancer cells. Intracellular concentration of drugs combined with heat shock causes impair of DNA repair-mechanism and correct DNA duplication. This action is magnified in tumor cells, that usually present an high proliferative index.
Hyperthermic isolated limb perfusion is indicated in following disease' stages:
- Palliative treatment for melanoma when residual life quality is the target.
- Curative ends in treatment of soft tissue sarcomas
- Limb saving procedure to avoid limb amputation
- Neoadiuvant treatment to reduce bulky tumors and allow successive conservative surgery.
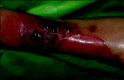
Figure 2
Lower limb after limb perfusion.

Figure 3
Same limb of Fig. 2 after 30 days from perfusion.

Figure 4
Same limb of Fig. 2 after 60 days from perfusion.
Surgical Procedure
Demolitive Phase
Lower Limbs
The iliac access is to be preferred for lower limbs. This access allows both iliac lymph nodes dissection and inguinal lymph nodes perfusion. Iliac nodes dissection is mandatory because in a non negligible percentage of patients (30% in some studies) an iliac-obturator lymph nodes disease can be present although the inguinal lymphonodes are uninjured. In some selected cases (second perfusion or previous iliac lymphadenectomy) femoral access has to be done. The iliac access requests the external iliac and obturator lymph nodes dissection with isolation of internal and external iliac vessels. The iliac perfusion, when groin lymphatic metastases are present, requires an inguenal-crural and iliac-obturator nodes en-bloc dissection. In this occurrence, a wide incision must be done, with section and reconstruction of inguinal ligament. This perfusion access is burdened by an high morbility (diastasis of wound margins, lymphorrea, wound infection). Recently, to reduce this morbility, we use to combine a laparoscopic iliac obturatory nodes dissection, associated to an “open” crural dissection. After this procedure we cannulate femoral vessels for perfusion. Going back to iliac access, when iliac vessels are isolated, we proceed towards femoral vessels, with isolation, ligature and section of circumflex, epigastric and other main collateral vessels. This manoeuvre reduces the drug leakage towards systemic circulation. In fact leakage is responsible both for systemic toxic effects and ineffective treatment, due to systemic hemodilution of the drug. After the isolation of collateral ramifications of iliac vessels, the radical iliac vessels nodes dissection is performed until the common iliac bifurcation (Fig. 5). Then follows the isolation on elastic string of obturator nerve, to preserve it from iatrogenic lesions, and obturator fossa dissection (Figs. 6, 7, 8). In our experience, lymphadenectomy is easier if performed with electrosurgical knife with bipolar forceps; moreover, the application of metallic clips makes safer the hemostasis and postoperative control of lymphorrhea due to lymph nodes dissection.

Figure 5
Iliac vessels after lymph nodes dissection.

Figure 6
Obturator nerve isolated on elastic string (blue string).

Figure 7
Obturator fossa nodes dissection.

Figure 8
Obturator nodes dissection completed.
When iliac access is not feasable or indicated, femoral access is done. We perform a longitudinal lozenge incision 2 cm medial and 2 cm caudal to the iliac anterior superior spine, that extends caudally to the apex of Scarpa's triangle. Follows a subcutaneous flap 1 cm in thickness to reach muscular sheath which is removed en bloc with subcutaneous flap, lymph nodes and perivascular tissue. During the preparation of the sheath and the lymphadenectomy, the safena vein, the deep circumflex, the epigastric vessels are tied up and dissected.
Upper Limbs
Patients is positioned with abducted limb, we perform an incision from sternoclavicular joint to the insertion of large pectoral muscle on humerus, then we lance the large pectoral sheath and divaricate the muscle fibers to reach the cephalic vein, that is tied up at its origin. The following step is small pectoral muscle section. After this phase, we isolate and tie up collateral vessels of axillary artery and vein to reduce the drug leakage. When vessels are isolated, the nodes dissection is performed.
Perfusion Time
During the preparatory phase, different temperature probes are positioned on limb (skin, tumor site, adductor muscles, pretibial muscles) for monitoring the temperature of whole limb during perfusion. To keep temperature uniformity along the treated region, the limb is covered with a double-sheet drape linked to an air-heater which is activated at the beginning of the operation (Fig. 9), When the complete iliac-obturator dissection has been obtained, we isolate on a tourniquet the internal and external iliac vessels. If internal vein isolation is difficult or dangerous we use to isolate the common iliac vein. When vessels are completely isolated and prepared for cannulation, we proceed to patient's heparinization (300 u.i. of heparin/kg), then we clamp external and internal iliac vessels (or common iliac vein) by tourniquets. On external vessels we perform an incision and place vascular catheters into iliac vessels. The position of catheters is very important: the extremities of the catheters must be placed before the bifurcation of common femoral vessels, just under the inguinal ligament (Fig. 10), In case of femoral access, femoral vessels are cannulated with the most cranial incision that can be done. The catheters are linked to the extracorporeal circulation system, consisting in an oxygenator, a heat exchanger, a peristaltic pump. The extracorporeal circulation starts at a minimum flow, that gradually increases until it reaches the appropriate flow to limb volumetry (usually a flow between 50 and 70 ml/liter of limb). After some minutes of circulation, the superficial blood circulation of limb is blocked by a rubber bandage twisted around the root of the limb and anchored to operative bed (Fig. 11).

Figure 9
Operatory room, limb heater linked to double sheet drape.

Figure 10
Cannulation of iliac vessels.

Figure 11
During perfusion, rubber tourniquet on the root of the limb.
When circulation is started, an important target to be pursuit is a constant level of solution inside oxygenator; this level is directly related to a balance between systemic and isolated limb circulation. This is accomplished equilibrating arterial and venous flow. To tell the truth, venous flow is determined from gravity, so the critical flow that we can regulate is arterial flow. All these contrivances, combined with ligation of collateral vessels and proper balance between systemic circulation and limb flow of perfusion, allows to keep low the drug leakage. Heat is given both from heat exchanger and double-sheet drape wrapped around the limb until an homogeneous temperature of 41.5°C is reached. When we get optimal temperature and perfusion flow balance, we inject the drug (melphalan 10 mg/liter tissues for lower limb and 12 mg/liter for upper limb) into the circuit. The drug dose is subdivided in three parts, administered in successive periods of time with an interval of 5 minutes. Perfusion is kept for 60 minutes after drug injection.
Leakage Monitoring
During hyperthermic isolated limb perfusion, the control of drug leakage to systemic circulation is of vital importance in order to keep low systemic toxicity and maximum drug concentration on tumor site. We measure leakage infusing in ECC circuit radioactively labelled human serum albumine with 99mTc. The 99mTc-labelled albumin is traced on liver-placed Geiger-Muller counter. Systemic leakage is quantitatively expressed as a percentage, whereby 100% leak is considered to give a homogeneous distribution of the tracer in the body. (Fig. 12). We consider appropriate a value of leakage ≤ 5%. It is important not to overcome a thresold value of 10%.

Figure 12
Geiger-Muller counter for leakage measure, the detection probe is positioned on the projection of the liver on abdominal wall.
Reconstruction Time
Once perfusion phase is finished, the vessels are uncannulated and sutured. Therefore the heparinization is neutralized by protamine solfate. The hemostasis is finished and, two morbid drains are placed (Fig. 13). In case of iliac access, one of them is positioned near the vessels submitted to lymphadenectomy and the other in the subcutaneous space. In case of femoral access a trasposition of sartorius muscle on femoral vessels is performed.This procedure is obtained disjoining the cranial head of the muscle from its bone insertion and traspositioning it on to adjacent vessels. The transposed muscle is fixed on the inguenal ligament with non reasorbable threads. This surgical procedure offers a support to the subcutaneous tissue, a protection to the femoral vessels from dehiscence or infection of the surgical wound and, at least, preserves patient from inguenal hernia that may follow this kind of operation.

Figure 13
At the end of perfusion: iliac vessels sutured.
Clinical Experiences and Results
From 1995 to December 2004, 79 patients with in transit metastases from melanoma, and 12 with sarcoma have been treated with 91 limb perfusions; in fact, five patients affected from melanoma have been treated twice. The main features of the patients wereº:
- Median age 65 years (35-81 range)
- 69 patients were at stage III (26 on stage IIIa; 43 on stage IIIAB)
- 10 patients at stage IV
- 76 lower limbs; 3 upper limbs
- All patients, outside of five, were previously treated with surgical removal of in transit metastases and/or with lymph nodes dissection.
- On 22 cases, more than 10 in transit metastases were present.
The follow-up and statistical elaboration of results confirmed the effectiveness of isolated limb perfusion. No perioperative mortality has been registered.
Major morbidity as follows:
- Edema of perfused limb (not only a complication, but an indirect sign of well-done perfusion), more or less, in each patient treated.
- 8 cases of transitory leucopenia and platelet disorder.
- 1 case of retroperitoneal hemorrage treated with conservative approach.
Late complications have been a wound diastasis and a chronic ischemia damage.
Our results are synthesized in Table 1 and are comparable to those reported in literature. The median survival has been evaluated in 48 months for patients at stage III and in 43 months for stage III AB.
Table 1
Clinical results in melanoma.
It is remarkable that also for patients at stage III and IV that presented a partial response, we obtained an adequate control of bulky disease and a good quality of patient's life until the death, generally occurred for systemic disease spreading. About patient affected from sarcomas, 12 patients have been treated with isolated limb perfusion. There was no perioperative mortality and the side effects have been:
- edema of the limb
- leucopenia (1 case)
- a compressive syndrome treated with a decompressive fasciotomy
For this kind of disease we have obtained an objective response on 80% of treated cases and we must underline that for almost all of them, the salvage of the limb has been possible, with improvement of life's quality.
References
- 1.
- Cavaliere R, Di Filippo F, Cavaliere F. et al. Medical radiology, thermoradiotherapy and thermochemotherapy. In: Seegenschmiedt MH, ed. Clinical Practice of Hyperthermic Extremity Perfusion in Combination with Radiotherapy and Chemotherapy. Springer-Verlag. 1996;2:323–345.
- 2.
- Krementz ET. Regional perfusion. Current sophistication, what next? Cancer. 1986;57:416. [PubMed: 3080216]
- 3.
- Schraffordt H, Koops BBR, Kroon FJ. et al. Management of local recurrence, satellites and in transit metastases of the limbs with isolation perfusion. In: Lejeune FJ, ed. Malignant Melanoma. McGraw-Hill. 1994:221–231.
- 4.
- Hahn GC. Hyperthermia and cancer. New York: Plenum Press. 1982
- 5.
- McBride M, McMurtrey MJ, Copeland EM. Regional Chemotherapy by Isolation- Perfusion. International Advances in Surgical Oncology. 1978. pp. 1–9. [PubMed: 553035]
- 6.
- Autier P. Epidemiology of Melanoma. In: Lejeune FJ, ed. Malignant Melanoma. McGraw-Hill. 1994:1–7.
- 7.
- Fraker DL, Coit DG. Isolated perfusion of extremity tumor. In: Lotze MT, ed. Regional Therapy of Advanced Cancer. Philadelphia: Lippincott-Raven. 1997:333–349.
- 8.
- Cavaliere F, Di Filippo, Cavaliere F. et al. Clinical practice of hyperthermic extremity perfusion in combination with radiotherapy and chemotherapy. In: Seegenschmiedt MH, ed. Medical Radiology, Thermoradiotherapy and Termochemotherapy. Springer-Verlag. 1996;2:323–345.
- 9.
- Clark AJ, Grabs PG, Parsons et al. Melphalan uptake, hyperthermic synergism and drug resistance in a human cell culture model for the isolated limb perfusion of melanoma. Melanoma Res. 1994;4(6):365–370. [PubMed: 7703715]
- 10.
- Di Filippo, Calabro A, Giannarelli D. et al. Prognostic variables in recurrent limb melanoma treated with hyperthermic antiblastic perfusion. Cancer. 1989;63:2551–2561. [PubMed: 2720604]
- 11.
- Bowers J, Copeland EM. Surgical limb perfusion for extremity melanoma. Surgical Oncology. 1994;3:91–102. [PubMed: 7952396]
- PubMedLinks to PubMed
- Hyperthermic Isolated Limb Perfusion - Madame Curie Bioscience DatabaseHyperthermic Isolated Limb Perfusion - Madame Curie Bioscience Database
- GBE1 Adult Polyglucosan Body Disease - GeneReviews®GBE1 Adult Polyglucosan Body Disease - GeneReviews®
- FRMD7-Related Infantile Nystagmus - GeneReviews®FRMD7-Related Infantile Nystagmus - GeneReviews®
- Congenital Stromal Corneal Dystrophy - GeneReviews®Congenital Stromal Corneal Dystrophy - GeneReviews®
- Phosphoribosylpyrophosphate Synthetase Deficiency - GeneReviews®Phosphoribosylpyrophosphate Synthetase Deficiency - GeneReviews®
Your browsing activity is empty.
Activity recording is turned off.
See more...
