NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Menini A, editor. The Neurobiology of Olfaction. Boca Raton (FL): CRC Press/Taylor & Francis; 2010.
One of the most characteristic features of odor memory in humans is the rather unique ability of odors to vividly trigger the evocation of emotional experiences. This property might be sustained by the direct connections established by the olfactory bulb and piriform/olfactory cortex on two structures involved in emotion and memory, namely the amygdala and hippocampus. In animals, memory for environmental odors plays a vital role because it regulates many behaviors that are crucial for survival. Highly emotional or at least particularly ethologically relevant olfactory learning occur during an animal’s major life events. Moreover, conditioning procedures can be used to induce emotional olfactory learning, thus allowing an experimental approach in a laboratory environment. The aim of the present chapter is to illustrate some aspects of the neurobiology of odor emotional memory in rats, both in infancy and adulthood. We focus on fear conditioning at both developmental points, since it constitutes one of the most adapted and classical paradigms to study emotional memory in animals and has been the focus of intense investigation. We begin by reviewing the available literature describing the neural circuits involved in odor fear conditioning in adult rats, and conclude the chapter by reviewing the ontogeny of odor fear conditioning. The same experimental paradigm can be supported by overlapping but distinct neural circuits, and lead to dramatically different behavioral outcome, depending on the age at learning. This approach highlights how the fear system changes to meet the ecological demands of different life stages.
15.1. INTRODUCTION
Although olfaction is generally assumed to play a minor role in humans compared to vision or audition, it is also commonly accepted that odors have a rather unique status for eliciting memories. Indeed, one of the most striking features of odor memory in humans resides in the amazing power of odors to vividly trigger the evocation of autobiographical experiences. This property has been referred to as the “Proust phenomenon” by some authors (Chu and Downes 2000, 2002) because of the well-known literary anecdote reported by Proust (1919) at the beginning of his novel, Swann’s Way, wherein the flavor of a madeleine cake dipped into a cup of tea unwillingly caused the remembrance of an old detailed memory.
“The memory suddenly appears before my mind. The taste was that of the little piece of madeleine which on Sunday mornings at Combray (because on those mornings I did not go out before church-time) my aunt Leonie used to give to me, dipping it first in her own cup of real or lime-flower tea’, leading him to the conclusion that ’When from a long-distant past nothing subsists … the smell and taste of things remain poised for a long time … and bear unfaltering, in the tiny and almost impalpable drop of their essence, the vast structure of recollection.”
A number of psychological studies have experimentally investigated the so-called Proust phenomenon in human subjects. Different works have shown that odor-cued memories are more emotional than memories triggered by visual or verbal cues (Chu and Downes 2000; Herz and Cupchik 1995; Herz 1998; Herz and Schooler 2002; Willander and Larsson 2007). Odor-cued memories have also been described as more vivid than memories evoked by corresponding words (Chu and Downes 2002). In addition, the feeling of being brought back in time to the occurrence of the event is experienced as stronger for odor-cued memories than memories evoked by words and pictures (Herz and Schooler 2002; Willander and Larsson 2006). Interestingly, it was also shown that autobiographical memories evoked by olfactory information were older than memories associated with verbal information (Chu and Downes 2000; Willander and Larsson 2006). Specifically, most odor-cued memories were located to the first decade of life (<10 years), whereas memories associated with verbal and visual cues peaked in early adulthood (11–20 years). This observation is in agreement with the fact that olfaction emerges very early in ontogeny and with research showing that associative odor learning begins very early in life (Schaal et al. 2000; Van Toller and Kendal-Reed 1995). Taken together, these studies suggest that human olfaction is unique in its ability to cue the emotional aspects of autobiographical memory, including experiences formed early in life.
In animals, memory for environmental odors plays a vital role because it regulates many behaviors that are crucial for their survival. Highly emotional or at least particularly ethologically relevant olfactory learning occurs during an animal’s major life events. For instance, and as will be developed in Section 15.3, during the early postnatal (PN) period when altricial pups are crucially dependent on their mother for feeding and maternal care, unusually rapid and strong olfactory learning occurs. The newly learned odor becomes preferred and is approached to bring the infant in contact with the mother, although the odor also controls the complex sequencing of motor patterns to support nipple attachment in rats (Cheslock et al. 2000; Hofer and Sullivan 2001; Pedersen et al. 1982) and rabbits (Distel and Hudson 1985; Coureaud et al. 2006; Schaal et al. 2003). At adulthood, two types of powerful ethological olfactory learning have been described in the literature. The first occurs in the postpartum period, when the mother learns the odor of the newborn and is critical for the mother to accept and nurse the young. This has been eloquently described in the postpartum sheep (Levy et al. 2004; Nowak et al. 2007; Keller et al. 2004, 2005; Brennan and Kendrick 2006) and rat (Fleming et al. 1999; Levy et al. 2004), and is the first step in the establishment of mother-young bonding. The second example of ecologically significant odor learning involves chemosensory individuality (Brennan 2004) and mate recognition (Brennan and Keverne 1997), which regulate reproductive behavior in mice (see also Chapter 6). Thus, particularly robust and rapid odor learning occurs throughout the life span during life transitions critical for survival and reproductive success.
Rapid, emotional learning is also necessary during threatening situations, to avoid that danger in the future. This type of learning is modeled by fear conditioning, where a temporal association of a neutral stimulus (i.e., tone or odor) is made with a noxious stimulus (i.e., shock) after only a few pairings. The formerly neutral stimulus acquires the ability to elicit defensive or escape responses critical in avoiding the danger and is dependent upon plasticity in the basolateral complex of the amygdala (LeDoux 2000; Fanselow and Gale 2003; Davis and Whalen 2001; Maren 2003; Otto et al. 2000). This paradigm has permitted enormous progress in our understanding of emotional learning in a controlled laboratory environment. More recently, this approach has been extended to humans with remarkable convergence in the underlying neural circuitry during acquisition, expression, and extinction, suggesting some homologous control of emotional learning in rats and humans (LaBar et al. 1998).
The aim of the present chapter is not to present an exhaustive review of the neurobiology of these different olfactory learning tasks, but rather to concentrate on those inducing emotional olfactory memory in rats, both at early PN ages and at adulthood. The vast majority of the existing studies on fear conditioning have been carried out using auditory or visual modalities. However, odors are at least as potent as auditory or visual cues in sustaining learned fear. Therefore, we specifically review the literature describing the neural circuit involved in odor fear conditioning in adult and infant rats, but we begin by presenting a brief review of the olfactory pathways within the brain.
15.1.1. Neuroanatomy of the Olfactory Pathways
Olfactory sensory neurons lie in the olfactory epithelium, which covers the back of the nasal cavity. The axons of the olfactory sensory neurons travel through the cribriform plate to the olfactory bulb, which is the first relay of olfactory information (Figure 15.1). The olfactory bulb output neurons send direct projections to the olfactory cortex, including the anterior olfactory nucleus, the olfactory tubercle, and the piriform or olfactory cortex (Shipley and Ennis 1996; Haberly and Price 1977; Carmichael et al. 1994). The first unusual characteristic of the mammalian olfactory pathways is that, unlike other sensory systems, it does not need to pass through the thalamus to reach cortical areas. Indeed, there is no thalamic relay between the first relay of sensory information, namely the olfactory bulb, and the primary olfactory cortex, as is the case for other sensory modalities (Figure 15.1). Similarly, there is no obligatory thalamic relay for olfactory information to reach the orbitofrontal cortex. Indeed, although the piriform cortex sends sparse projections to the mediodorsal thalamus, thereby disynaptically targeting the orbitofrontal cortex, it establishes direct monosynaptic connections with the orbitofrontal cortex (Datiche and Cattarelli 1996; Tanabe et al. 1975; Yarita et al. 1980; Ray and Price 1992; Carmichael et al. 1994). These observations suggest that the thalamus might be less relevant for olfaction than for the other senses (Shepherd 2005; Wilson and Stevenson 2006), but see Plailly et al. (2008) for an alternative view.
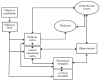
FIGURE 15.1
Schematic representation of the main components of the olfactory pathways. Diagram illustrates the intimate link between the olfactory bulb and the limbic areas (amygdala and hippocampus).
Another unique feature of the olfactory pathways is the olfactory bulb’s output neurons rapid connections to structures crucially implicated in emotion and memory, namely the amygdala and the hippocampus. Indeed, the main olfactory bulb makes dense monosynaptic contacts with nuclei of the corticomedial amygdaloid group, including the nucleus of the lateral olfactory tract, the cortical nucleus of the amygdala, and the periamygdaloid cortex (Price 1973; McDonald 1998). These observations led Swanson and Petrovich (1998) to suggest that the corticomedial amygdala is an integral component of the olfactory system. These superficial nuclei are a major source of the projections from the amygdala to the hypothalamus (Price et al. 1991). In contrast, the deeper amygdaloid nuclei, including the basolateral nuclear group, do not receive projections from the olfactory bulb and receive relatively weak projections from the olfactory piriform cortex (Krettek and Price 1978; Luskin and Price 1983; Ottersen 1982). However, they receive fairly dense projections from the corticomedial nuclei within the amygdala (Savander et al. 1996). Taken together, these anatomical data suggest that compared to the other sensory modalities, olfactory information has a unique direct access to the amygdala.
In addition, both anatomical (Price 1973; Haberly and Price 1977) and electrophysiological (Biella and de Curtis 2000; Wilson and Steward 1978; Boeijinga and Van Groen 1984; Mouly et al. 1998, 2001; Mouly and Di Scala 2006; Biella et al. 2003) studies have shown that the olfactory bulb and the piriform cortex also send direct projections to the lateral entorhinal cortex via the lateral olfactory tract. The lateral entorhinal cortex, in turn, has been shown to project to the hippocampus via the lateral perforant path (Witter and Amaral 1991). Furthermore, in addition to providing a gateway to the hippocampus, the lateral entorhinal cortex sends monosynaptic projections to the amygdala. The primary targets of the entorhinal cortex are the basolateral amygdala and the cortical nuclear complex (Brothers and Finch 1985; McDonald and Mascagni 1997). The lateral entorhinal cortex is, therefore, in a position to transmit information from olfactory cortical areas to two major telencephalic components, the amygdala and the hippocampus, and back to olfactory areas through reciprocal connections (Swanson and Kohler 1986; Insausti et al. 1997).
15.1.2. Ontogenesis of the Olfactory Pathways
The olfactory system is functional at birth and, as outlined in Section 15.3, odors support behaviors critical for pups’ survival at birth. Amazingly, pups smell, discriminate, and process odors, yet the olfactory circuit of pups is quite immature compared to the adult rat brain. While the olfactory bulb is fairly mature at birth and functional odor maps in the glomerular layer are well defined, considerable development continues throughout the preweanling period (Guthrie and Gall 1995, 2003). For example, gamma-aminobutyric acid (GABA) interneurons, presumably critical for odor discrimination in adults (Ennis et al. 1998), are sparse, yet pups show odor discrimination (Fletcher et al. 2005). Additionally, centrifugal input from the rest of the brain begins to enter the bulb prenatally, although substantial input continues through weaning. For example, norepinephrine (NE) fibers from the locus coeruleus (LC) are present at birth, although NE released by these terminals greatly decreases as pups mature (McLean and Shipley 1991), while terminal numbers increase. While minor cholinergic (ACh) projection neurons arrive in the bulb as early as the first day of life to innervate the modified glomerular complex, most fibers begin to reach the bulb at PN day 6 and start to show the more typical homogenous distribution across the bulb layers (McLean and Shipley 1991; Le Jeune and Jourdan 1991). At birth, there is also a transient large number of neurons expressing acetylcholinesterase (AChE), although this begins to decrease around PN15, and shows low adult levels by PN30 (Le Jeune and Jourdan 1991). In the next relay station for olfactory information, the olfactory cortex, considerable development also occurs during the first two weeks of life and later (Brunjes et al. 2005; Schwob and Price 1984; Walz et al. 2006). However, the functional significance of the developing piriform on pup behavior has remained elusive, primarily due to lack of research attention.
The amygdala also continues to develop during the period. The basolateral complex first emerges at E17, with most neurons produced between E20-PN7. Other amygdaloid nuclei lag behind the basolateral by a few days. The amygdala continues to develop until adolescence (Morys et al. 1998, 1999; Berdel and Morys 2000; Dziewiatkowski et al. 1998; Berdel et al. 1997). Olfactory information is received by the amygdala in the early neonatal period (Schwob and Price 1984) and the infant amygdala responds to odors (Thompson et al. 2008). However, the sequential emergence of amygdala-dependent behaviors throughout the preweanling period suggests important intra-amygdala connections continue (Hunt et al. 1994). Functional maturation of the amygdala and its role in pups’ behavior is outlined in Section 15.3.
Connectivity between olfactory structures continues to become more complex. For example, in the infant rat, frontal cortex development and its projections to the piriform cortex and amygdala are also protracted, with anatomical maturation and connections occurring between PN8–14, although “adult-like” laminar cellular organization occurs as pups complete the second week of life (Bouwmeester et al. 2002; Verwer et al. 1996; Kolb et al. 1996). Together, the protracted development of these brain areas suggests that the neonate has a unique circuitry for odor processing that underlies developmental changes in emotional learning.
15.2. EMOTIONAL ODOR MEMORY IN ADULT RATS
15.2.1. Fear Conditioning: A Tool for Studying Emotional Memory in Animals
Among the few emotions that can be detected and quantified in animals, fear is indubitably the most common. When a rat encounters a threatening stimulus (e.g., the odor of a predator), it expresses fear reactions, including physiological (blood pressure increase, stress hormones release, hypoal-gesia, etc.) and behavioral responses (Takahashi et al. 2005; Maren and Quirk 2004; Dielenberg and McGregor 2001). Among these, fear-potentiated startle and fear-induced freezing (defined as complete immobility) are universal fear responses seen in many animal species, including humans. These innate fear responses to a natural threat can be classically conditioned to an experimental neutral stimulus and this is referred to as fear conditioning (LeDoux 2000; Maren 2001; Myers and Davis 2007; Maren and Quirk 2004). Fear conditioning consists of pairing an initially neutral stimulus (the conditioned stimulus or CS) with an aversive unconditioned stimulus (US), generally a mild foot-shock. After a few trials, re-exposure to the CS alone elicits fear responses, such as freezing behavior, assumed to be part of an anticipatory response to threat and danger (Rosen and Schulkin 1998).
The vast majority of studies devoted to investigate the neural basis of fear conditioning have used auditory and, to a lesser extent, visual cues as conditioned stimuli. In particular, the neural pathways involved in auditory fear conditioning have been well characterized (LeDoux 2000; Maren 2001; Maren and Quirk 2004; Myers and Davis 2007). The information carried by the auditory CS can take one of two pathways: either directly from the thalamus to quickly reach the amygdala or the CS can travel from the auditory thalamus to the auditory cortex before reaching the amygdala. These thalamic and cortical areas send -projections to the lateral nucleus of the amygdala, which is a site of CS-US convergence. The lateral nucleus, in turn, projects to the central amygdala, which controls the expression of fear responses by way of projections to brainstem areas. The major conclusion of these studies is that the amygdala plays a critical role in linking external stimuli to defense responses through synaptic plasticity. The amygdala would, therefore, be essential to the formation and storage of fear memories as far as auditory or visual stimuli are used for conditioning. Extinction of fear memory would involve an inhibitory action exerted by the prefrontal cortex onto the amygdala network (Milad and Quirk 2002), although disruption of memory within the amygdala has also been implicated (Davis et al. 2003). In addition, the hippocampus seems to play an important role in regulating the context dependence of fear memory after extinction.
Intriguingly, whereas olfaction plays a dominant role in rat’s behavior from the very first hours of life throughout adulthood and, as outlined above, olfactory areas establish particularly direct connections with the amygdala, very few studies have used odor cues as CS in fear conditioning paradigms.
15.2.2. Neurobiology of Olfactory Fear Conditioning in Rats
Behavioral researches have shown that olfactory fear conditioning induces robust emotional responses. Otto et al. (1997, 2000), measuring freezing behavior as an index of learned fear, reported that olfactory fear conditioning resulted in robust and long-lasting associations between an odor and a foot-shock. Richardson et al. (1999) and later, Paschall and Davis (2002), using fear-potentiated startle as another measure of learned fear, showed that an odor previously paired with shock was a particularly effective stimulus for potentiating the startle response in rats. Moreover, in a subsequent study, Richardson et al. (2002) reported that extinction of learned fear potentiation of startle occurs more slowly with an olfactory CS than with auditory or visual cues. Thus, odors are at least as efficient as other types of sensory stimuli to induce learned fear responses.
15.2.2.1. The Role of the Amygdala
Concerning the neural basis of olfactory fear conditioning, there is a consensus in the existing literature that the amygdala plays a crucial role. Indeed, Cousens and Otto (1998) first showed that pre- and posttraining excitotoxic lesions of the basolateral amygdala abolished the acquisition and expression of olfactory fear conditioning in rats. In another study, Walker et al. (2005) evaluated the respective contribution of the basolateral and medial amygdala to olfactory fear conditioning by infusing antagonists of glutamate receptors into these areas prior to either odor-shock pairings or retention test using fear-potentiated startle. Pretraining blockade of N-methyl D-aspartate (NMDA) receptors into the basolateral amygdala disrupted fear conditioning to the odor, while pretest blockade of α-amino-3-hydroxyl-5-methyl-4-isoxazole-propionate (AMPA)/kainate receptors disrupted fear-potentiated startle to the learned odor. Neither compound blocked fear conditioning when infused into the medial amygdala prior to training, but pretest AMPA/kainate receptors antagonist infusion did block fear-potentiated startle. Using an electrophysiological approach, Rosenkranz and Grace (2002) performed in vivo intracellular recordings in the lateral nucleus of the basolateral amygdala during acquisition of an odor fear conditioning under anaesthesia, in rats. Their data revealed that repeated pairing of an odor with foot-shock induced enhanced postsynaptic potential responses in neurons of the basolateral amygdala, and that this modification resulted from local changes in synaptic efficacy, and was dependant on dopamine.
Another set of studies has investigated more specifically the involvement of the amygdala in the consolidation of odor fear conditioning. Specifically, Killpatrick and Cahill (2003) used reversible inactivation of the basolateral amygdala with tetrodotoxin following paired odor-shock presentation in rats. This treatment resulted in a deficit in learning, thus suggesting that the basolateral amygdala plays a role in consolidation of odor fear conditioning. Similarly, Rattiner et al. (2004) measured the expression of brain-derived neurotrophic factor (BDNF) mRNA in the amygdala after olfactory as well as visual fear conditioning. BDNF is known to play a critical role in activity-dependent synaptic plasticity and has been implicated as mediator of hippocampal-dependent learning and memory (Hall et al. 2000; Kesslak et al. 1998). The authors showed that BDNF expression was increased in the basolateral amygdala two hours after fear conditioning and that this increase occurs independently of the sensory modality of the CS.
Taken together, these data suggest that the basolateral amygdala plays a major role in the acquisition, consolidation, and retention of olfactory fear conditioning, thus extending to odor cues the previous observations obtained with auditory and visual CS.
15.2.2.2. The Role of Extra-Amygdala Structures
Beside the above studies specifically addressing the role of the basolateral amygdala, sparse studies have investigated the potential involvement of other target structures in this learning. Among them, Ressler et al. (2002) used genes identified in a kainic acid model of synaptic plasticity, as in situ hybridization probes during the consolidation period after odor fear conditioning. They found that following odor-shock pairings, these genes (such as immediate early genes like c-Fos and Zif 268) were transcriptionally regulated in several brain areas, including the basolateral and medial amygdala, the piriform, perirhinal, and insular cortices, along with the endopiriform nucleus and the habenula. These data suggest that plasticity after odor fear conditioning may depend on a broad neural circuit that includes the amygdala.
Other studies have investigated more precisely the role of extra-amygdala areas in odor fear conditioning. Laviolette et al. (2005) examined the possible role of the medial prefrontal cortex (mPFC) in the acquisition and encoding of odor fear learning at the behavioral and single-neuron level. A subpopulation of neurons in the mPFC that received monosynaptic inputs from the basolateral amygdala demonstrated strong associative responding to odors paired previously with foot-shock by increasing spontaneous activity and bursting activity. In addition, systemic or intra-mPFC blockade of dopamine receptors prevented this emotional associative learning in neurons of the mPFC and blocked the expression of olfactory conditioned fear. These results demonstrate that mPFC neurons that receive a functional input from the basolateral amygdala actively encode emotional learning and that this process is under the dependence of dopamine.
In another study using aspirative or excitotoxic lesions, Herzog and Otto (1997, 1998) investigated the role of the perirhinal cortex. They reported that rats with perirhinal cortex lesions presented a robust attenuation of fear conditioning to olfactory stimulus, thus, suggesting that the perirhinal cortex is an important component of the neural circuit supporting the association between olfactory cues and foot-shock. Later, Schettino and Otto (2001), measuring c-Fos expression related to the acquisition and expression of olfactory fear conditioning, reported that the anterior region of the medial nucleus of the amygdala, as well as the ventral perirhinal cortex, could be critically involved in this learning. Using the same technique, Funk and Amir (2000) also showed that presentation of the aversive conditioned odor stimulus induced an enhanced increase in levels of Fos expression in the main and accessory olfactory bulbs and in the anterior olfactory nucleus, as well as in the infralimbic and orbital cortices.
15.2.2.2.1. The Role of the Hippocampus
In the literature, there is widespread agreement that the hippocampus is not critically involved in the acquisition of discrete CS-US associations (Anagnostaras et al. 2001; Lopez-Fernandez et al. 2007; Maren et al. 1997; Phillips and LeDoux 1992; Schafe et al. 2001), and this also stands for odor-shock pairings (Otto and Poon 2006). Interestingly however, when the odor is presented as a contextual cue instead of an elemental discrete cue, the hippocampus seems to be engaged in the learning of the association. Specifically, Parsons and Otto (2008) examined the effects of temporary inactivation of the dorsal hippocampus on fear behavior in an explicitly nonspatial contextual fear conditioning paradigm, in which olfactory stimuli served as temporally and spatially diffuse contexts. They found that the transient lesion of the dorsal hippocampus produced both anterograde and retrograde deficits in olfactory contextual conditioning, while sparing the acquisition and expression of freezing to a discrete auditory or olfactory CS. These data suggest that the dorsal hippocampus participates in both the acquisition and retention of unimodal olfactory contextual fear conditioning.
15.2.2.2.2. The Role of the Piriform Cortex
Among the other candidate structures for participating in the neural network sustaining odor fear learning, the piriform cortex has been the core of particular attention. The piriform cortex is the largest of the olfactory areas and is usually divided into anterior and posterior parts. Olfactory bulb output neurons synapse primarily on pyramidal cells in the anterior piriform cortex and only send lighter, more distributed inputs to the posterior piriform cortex (de Olmos et al. 1978; Haberly and Price 1978; Haberly 2001). Furthermore, the ratio of associative to afferent input is higher in more posterior regions of the piriform cortex (Haberly and Price 1978; Luskin and Price 1983; Datiche et al. 1996). Moreover, whereas the anterior piriform cortex receives input from the orbitofrontal cortex (Datiche and Cattarelli 1996; Illig 2005), the posterior piriform cortex receives afferents from the basolateral amygdala (Johnson et al. 2000; Majak et al. 2004). In addition, electrophysiological studies have further emphasized this dissociation. Indeed, Litaudon and Cattarelli (1996), using optical recording of the whole piriform cortex activity, showed that following repetitive electrical stimulation of the olfactory bulb, a higher degree of inhibition was observed in the ventromedial anterior part of the piriform cortex than in the posterior part. Using the same technique, Litaudon and Cattarelli (1995) also found that the amplitude of a late component of the evoked activity was larger in the posterior than in the anterior piriform cortex, a result which was confirmed by Mouly et al. (1998) on classical evoked field potential signals. In the domain of synaptic plasticity, Jung et al. (1990) showed that the induction of long-term potentiation in vitro was easier in posterior piriform cortex slices than in the anterior slices. Taken together, the anatomical and functional differences observed between anterior and posterior piriform cortices suggest that these two parts of the olfactory cortex could play differential roles in odor fear conditioning.
Jones et al. (2007) examined BDNF mRNA expression across the olfactory system following fear conditioning in mice. They found a specific increase in BDNF mRNA in animals receiving paired foot-shocks in the posterior piriform cortex and the basolateral amygdala. This was in contrast to the unpaired and odor-alone treatments, where BDNF mRNA was increased in the olfactory bulb and the anterior piriform cortex only, but not the higher olfactory areas. Thus, BDNF mRNA in the olfactory bulb and the anterior piriform cortex seems to be induced simply by exposure to olfactory stimuli, regardless of whether the odor is associated with the shock. In contrast, BDNF mRNA is only induced in the posterior piriform cortex and the basolateral amygdala when there is an association between odor and shock to support learning.
These data complement previous observations reported in our group. Indeed, using an electrophysiological approach, we investigated whether olfactory fear conditioning induces synaptic changes within the olfactory pathways, mainly focusing on olfactory cortical areas, namely the piriform cortex and the amygdala cortical and basolateral nuclei (Sevelinges et al. 2004). For this, evoked field potential signals induced in behaving animals by electrical stimulation of the olfactory bulb were collected simultaneously at the level of the anterior piriform cortex, the posterior piriform cortex, the cortical nucleus of the amygdala, and the basolateral nucleus of the amygdala. Recordings were made before learning and during a retention test carried out 24 h after acquisition of odor fear conditioning. The data showed that learning was accompanied by a lasting increase in signal amplitude in the cortical amygdala, i.e., an increase observed before CS presentation (Figure 15.2A). In addition, introduction of the learned CS odor induced a transient facilitation in the basolateral amygdala, as well as in the cortical amygdala and the posterior piriform cortex (Figure 15.2B). These data suggest that the posterior piriform cortex and the amygdala are key structures in the neural circuit underlying odor fear conditioning.
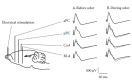
FIGURE 15.2
Evoked field potentials were induced in the anterior piriform cortex (aPC), the posterior piriform cortex (pPC), the cortical (CoA) and basolateral nucleus of the amygdala (BLA) in response to stimulation of the olfactory bulb, either before (plain line) (more...)
In a recent study (Hegoburu et al. In preparation), we used high temporal resolution (1 min sampling rate) intracerebral microdialysis, in order to study the accurate temporal dynamic of neurotransmitter release in these two structures during the course of learning acquisition. Simultaneous monitoring of GABA and glutamate was performed in both the basolateral amygdala and the posterior piriform cortex, during the odor fear acquisition session including six odor-shock pairings. A transient increase in glutamate was observed in the amygdala for the two first CS-US pairings, whereas a significant increase in GABA levels was shown for the second pairing only. In contrast, in the posterior piriform cortex, transient increases in both GABA and glutamate levels were found after each odor-shock pairing. Interestingly, for both neurotransmitters, the increase observed in the amygdala occurred earlier in time (1–2 min) than the increase in the posterior piriform cortex. High temporal resolution microdialysis, therefore, allowed us to show a temporal dynamic of activation of these two structures during the course of successive pairings, with the involvement of the amygdala during the first odor-shock associations preceding neurotransmitter release in the posterior piriform cortex, after which the latter alone supports pairing-induced modifications. This led us to suggest that the amygdala has a crucial, but temporally limited, role in signaling changes during a potentially dangerous situation, while the posterior piriform cortex may contribute to higher mnemonic processes, including storage of the detailed attributes of the learned stimulus
Taken together, these data bring further support to the growing body of evidence showing that the anterior and posterior piriform cortices play different complementary roles in memory processes. Specifically, the anterior piriform cortex would be mainly involved in coding the sensory features of the odor (Roesch et al. 2007), and in simple forms of short-term memory like habituation (Kadohisa and Wilson 2006; Wilson 1998, 2000) or perceptual learning (Barnes et al. 2008; Wilson and Stevenson 2003), whereas the posterior piriform cortex would be involved in the learning and recall of associations between odorants and information from other sensory modalities (Chabaud et al. 1999, 2000; Mouly et al. 2001; Mouly and Gervais 2002; Kadohisa and Wilson 2006; Haberly 2001; Litaudon et al. 2003; Sevelinges et al. 2004, 2008; Calu et al. 2007; Martin et al. 2004).
15.2.3. Odor Fear Conditioning in Humans
During the past ten years, several studies using the functional magnetic resonance imaging (fMRI) technique, have investigated the networks involved in fear conditioning in humans (Cheng et al. 2003; Buchel and Dolan 2000; LaBar et al. 1998). Most of these studies suggest that the amygdala is part of the circuit, thus corroborating the data from the animal literature. Interestingly, in a recent work, Li et al. (2008) used an odor fear conditioning paradigm in humans in order to investigate how aversive learning enhances perceptual acuity of sensory signal. During conditioning, the CS odor presentation coterminated with electric shock (US), whereas presentation of its chiral counterpart (enantiomer) was not associated with the US. The authors combined multivariate fMRI with olfactory psychophysics, and hereby showed that initially indistinguishable odor enantiomers become discriminable after aversive conditioning. In parallel, fMRI data demonstrated progressive decreases in amygdala activity evoked by the learned aversive odor as learning proceeded, together with increases in the orbitofrontal cortex. Interestingly, the authors also measured changes in activity in the piriform cortex. They reported that spatial patterns of fMRI activity in the posterior piriform cortex between the two enantiomers were highly correlated before conditioning, but became more distinct after conditioning. This effect was specific to the posterior piriform cortex, as it was not observed in the anterior piriform cortex. These findings confirm and extend previous data reported by the same group, showing a double dissociation in the piriform cortex, whereby posterior regions encode quality, and anterior regions encode structure (Gottfried et al. 2006; Li et al. 2006). Taken together, these data indicate that aversive learning induces plasticity in the posterior piriform cortex that correlates with gains in odor enantiomer discrimination. This led the authors to propose that fear conditioning has the capacity to update perceptual representation of predictive cues, in addition to its well-recognized role in the acquisition of conditioned responses.
The data obtained in human odor fear conditioning are strikingly similar to those described in rats and suggest that whereas the amygdala plays a crucial role, a broad network of structures is involved in the learning, among which the piriform cortex seems to endorse a privileged status.
15.3. DEVELOPMENT OF ODOR LEARNING MEMORY
The importance of emotional olfactory learning is evident in the rat pups’ first learning experience, when pups must learn the mother’s odor to attach to their caregiver. However, pup learning is strikingly different from adult learning and appears to accommodate pups’ unique environmental demands. Pups are born immature and completely dependent upon their mother for survival, yet become independent only three weeks later. During this time, the rat pups’ world repeatedly changes as they transition from intrauterine life, to nest life, and finally to independence. As will be illustrated in this chapter, different forms of learning emerge, disappear, and sometimes coexist as pups mature. Here, we will review the relationship between a few forms of olfactory learning that change as the demands and importance of attachment learning to the mother and more “adultlike” learning interplay. We will review three types of infant learning: (1) attachment learning that involves learning the maternal odor and expressing the complex behaviors controlled by that odor; (2) olfactory fear learning that emerges as pups begin to venture outside the nest; and (3) malaise learning that enables pups to avoid odors paired with gastrointestinal distress, learning that occurs throughout development but with a changing neural circuit.
15.3.1. Attachment Learning and Maternal Odor
Perhaps the most dramatic transition occurs at birth when infant rats face the daunting cognitive task of transitioning from intrauterine life to nest life. Not only do pups need to learn their mother’s odor within minutes of birth, but also they must rapidly express this learning with approach to their mother and nipple attachment for their first nursing bout. While the mother does assist pups in maintaining contact with her, and prenatal learning supports the PN learning, the PN learning and expression are essential (Pedersen and Blass 1982; Polan and Hofer 1999). If pups do not learn the maternal odor or their olfactory function is disrupted, they have little chance of survival (Singh et al. 1976). Furthermore, since the maternal odor is dependent on the maternal diet, pups must continue to learn the maternal odor throughout the preweanling period.
The maternal odor guides pups to the mother and controls social interactions with the mother, including nipple attachment. During the first week and a half of life, this attachment learning shows very rapid and robust acquisition, as well as some intriguing unique characteristics similar to imprinting. This sensitive period for pups’ rapid learning appears well adapted for the unique demands of life within the nest and the transition to independence that occurs a few weeks later. For most of the sensitive period, the motorically immature pups remain in the nest nursing. However, the mother entering and leaving the nest is sometimes associated with distress to pups when the mother drags still-attached pups out of the nest and steps on others. Thus, pups’ exposure to the maternal odor occurs during nursing and grooming by the mother, but with occasional painful stimuli from the mother. Importantly, pups feel pain (Fitzgerald 2005; Collier and Bolles 1980; Barr 1995; Emerich et al. 1985; Shair et al. 1997; Stehouwer and Campbell 1978). As illustrated in Figure 15.3, one of the most strikingly unique learning characteristics of pups, is the ability of both painful stimuli (0.5 mA shock, tailpinch) and presumably pleasant stimuli (milk, warmth, stroking that mimics maternal grooming) to both support learning of approach responses and nipple attachment. Specifically, pairing a novel odor (CS) with either a painful or presumably pleasant reward, results in pups showing subsequent approach responses to that odor. Indeed, this learning occurs within the nest with a mother nursing her pups, but also when pain is induced by a stressed mother (Gilles et al. 1996), who exhibits rough handling and transporting of pups without nursing (Roth and Sullivan 2005). This attenuated odor-aversion or fear learning during pups’ early life is not the only learning restriction for pups, since inhibitory conditioning and passive avoidance are also attenuated (Blozovski and Cudennec 1980; Collier and Mast 1979). Finally, these early life learned odors are not just approached as a preferred odor, but appear to take on characteristics of maternal odor and also support nipple attachment when the natural maternal odor is removed (Pedersen and Blass 1982; Raineki et al. in progress; Hofer et al. 1976). Thus, this learning is unique both in its expression and acquisition and has been characterized as mammalian imprinting (Hofer and Sullivan 2001).

FIGURE 15.3
Y-maze test in sensitive-period PN8 pups using the CS odor or the maternal odor vs clean home cage bedding (response to bedding not shown). Infant rat pups can be classically conditioned to approach a novel odor with previous pairings with either painful (more...)
The early life odor learning that supports acquisition of the maternal odor is also potentiated by other unique learning features in infancy. Specifically, exposure to the odor CS before classical conditioning (latent inhibition) and uncorrelated presentations of the CS and reward (learned irrelevance), both of which retard/inhibit learning in adults, either enhance or have no effect on the young infant rat’s learning (Stanton 2000; Stanton et al. 1998; Campbell and Spear 1972; Hoffmann and Spear 1988; Rescorla 1967, 1988; Rush et al. 2001; Siegel and Domjan 1971; Spear and Rudy 1991). Finally, while sequential presentation of the CS and the reward optimizes learning in adults, simultaneous presentation of stimuli enhances associations in young pups (Barr et al. 2003; Cheslock et al. 2003).
Thus, rat pups have myriad unique learning characteristics that potentiate the odor learning required for pups to maintain proximity to their mother. While one normally thinks of early life learning as providing enhancement of learning, such as in imprinting, limitations on aversive learning in early life are actually widespread. For example, shocking a chick while it is following the surrogate caregiver during imprinting, enhances following of the surrogate caregiver. Just hours after the imprinting critical period ends, a similar shocking procedure causes the chick to avoid the surrogate (Salzen 1967). An analogous situation has been demonstrated in young dogs (Stanley 1962). Indeed, mistreating a puppy with shock or rough handling by a human caregiver results in a strong attachment to that caregiver. A similar phenomenon has been repeatedly documented in nonhuman primate colonies, where the young of abusive mothers still form and maintain strong attachments to their caregiver (Harlow and Harlow 1965; Maestripieri et al. 1999; Sanchez et al. 2001; Suomi 2003; Nemeroff 2004). Finally, clinical and epidemiological studies on children indicate that “abuse” results in children forming and maintaining strong attachments to their caregiver (Bowlby 1965; Helfer et al. 1997). Indeed, the wide phylogenetic representation of tolerance of pain during attachment learning and its maintenance suggests a phylogenetically preserved system for attachment (Hofer and Sullivan 2008).
15.3.2. The Neurobiology of Attachment Odor Learning: Olfactory Bulb, Anterior Piriform Cortex, and Locus Coeruleus (LC)
During the sensitive period, the neural circuitry supporting the attachment olfactory learning and memory also appears unique and is associated with enhanced odor-induced olfactory responding. Similarly to the behavior, the neural responses evoked by natural maternal odors and attachment odors learned within the nest or in controlled learning experiments appear similar (Harley et al. 2006; Sullivan et al. 1990; McLean and Shipley 1991; Woo et al. 1996; Roth and Sullivan 2005; Shionoya et al. 2006; Leon and Johnson 2003; Yuan et al. 2000; Pedersen and Blass 1982; Smotherman 1982). This modified olfactory bulb response has been documented using 2-DG, c-Fos, intrinsic optical imaging, and modified single-unit response patterns of the bulb’s output neurons, mitral/tufted cells (Johnson et al. 1995; Sullivan and Leon 1986; McLean and Shipley 1991; Wilson et al. 1987; Woo et al. 1987). Recent work also suggests that this early life odor preference learning supporting attachment is also encoded in the anterior piriform cortex (Moriceau and Sullivan 2006; Roth and Sullivan 2005). Due to limited research on the immature piriform cortex, its function in pup learning remains unknown. As reviewed above, both the anterior and posterior piriform cortices have an important yet different role in adult odor learning (Sevelinges et al. 2004; Kadohisa and Wilson 2006; Calu et al. 2007; Brosh et al. 2006; Brennan and Kendrick 2006; Martin et al. 2004, 2006; Bernabeu et al. 2006; Datiche et al. 2001).
During the sensitive period, the olfactory attachment odor and the olfactory bulb learning-induced changes are dependent upon high levels of NE (Sullivan and Wilson 1994; Harley et al. 2006; Sullivan et al. 2000), with the LC as the bulb’s sole source of NE (McLean and Shipley 1991). The sensitive-period LC is uniquely responsive to diverse types of sensory stimulation (e.g., 1 s stroking, 0.5 mA shock), which may account for the wide range of sensory stimuli to support infant learning. Additionally, the sensitive-period LC releases abundant NE (Rangel and Leon 1995; Moriceau et al. In preparation) due to its prolonged response (20–30 s), which is in sharp contrast to the very brief millisecond response found in older pups and adults (for a review, see Nakamura and Sakaguchi 1990). Finally, the infant LC fails to habituate with repeated stimulus presentations, while the adult LC rapidly habituates with only two or three presentations (Nakamura and Sakaguchi 1990; Nakamura et al. 1988; Sara et al. 1994).
With maturation (>PN10), NE release from the LC is greatly reduced and no longer sufficient to produce odor preference learning in postsensitive-period pups (Sullivan and Wilson 1994; Moriceau and Sullivan 2004; Sullivan et al. 2000; Harley et al. 2006). This changing developmental role for LC and its reduced NE release is due to the functional emergence of the LC’s inhibitory αc2 noradrenergic autoreceptors that quickly terminate the LC’s response and greatly reduces NE release (Nakamura and Sakaguchi 1990; McGaugh 2006). Interestingly, the olfactory bulb’s ability to support odor attachment learning can be reinstated by direct infusion of high levels of NE or by blocking the LC’s α2 noradrenergic autoreceptors to reinstate the LC’s prolonged response and abundant NE release (Moriceau and Sullivan 2004). With this postsensitive-period LC maturation, NE begins to play a more modulatory role of enhancing or attenuating memories in a manner similar to adults (for reviews, see McGaugh 2006; Ferry and McGaugh 2000). Thus, while many neurotransmitters have a role in early olfactory learning in neonatal rats, NE appears to have a particularly important role.
15.3.3. Emergence of Olfactory Fear Learning
At around PN10, crawling transition to walking, and the probability of pups leaving the nest increases. Pup odor attachment learning is not appropriate for outside the nest, where learning needs to reflect the consequences of pain or pleasure to ensure pups survival in preparation for independent life. Indeed, the sensitive-period attachment learning ends at this time, at least when pups are outside the nest. Furthermore, new learning emerges and pups begin to exhibit olfactory fear conditioning from odor-pain pairings (0.5 mA shock, tailpinch), with pups avoiding the odor in a two-odor choice Y-maze and freezing to CS odor presentation in a glass jar (Roth and Sullivan 2005; Haroutunian and Campbell 1979; Moriceau and Sullivan 2006; Moriceau et al. 2006; Sullivan and Leon 1986; Sullivan et al. 2000) (Figure 15.4).

FIGURE 15.4
The graphic on the left illustrates the neural basis of the attachment learning that occurs when an odor is paired with myriad stimuli (stroking, warmth, milk, 0.5 mA shock). Without the mother present, this learning occurs in pups younger than PN10. (more...)
15.3.4. The Neural Basis of Fear Conditioning the Piriform Cortex and Amygdala
The amygdala is required for fear conditioning (Blair et al. 2001; Sananes and Campbell 1989; Fanselow and LeDoux 1999; Davis et al. 1997; Fanselow and Gale 2003; Herzog and Otto 1997; Maren 2003; Pape and Stork 2003; Pare et al. 2004; Rosenkranz and Grace 2002; Schettino and Otto 2001; Sevelinges et al. 2004; Sigurdsson et al. 2007; Davis et al. 2003). Indeed, as fear conditioning emerges in pups, the basolateral complex of the amygdala shows learning-associated changes, as indicated by c-Fos and 2-DG (Moriceau and Sullivan 2006; Moriceau et al. 2006; Sullivan et al. 2000). Importantly, a causal relationship has been established between the currently emerging fear conditioning and the amygdala, since temporarily silencing the amygdala with muscimol abolishes pups fear conditioning (Moriceau and Sullivan 2006).
Input to the amygdala also changes at this age. Before the emergence of the amygdala-dependent fear conditioning, the olfactory bulb and anterior piriform were associated with the odor-shock induced odor preference (Moriceau et al. 2006; Roth and Sullivan 2005). The olfactory bulb appears to indicate an odor is important with the hedonic value coded by the anterior piriform cortex (Moriceau et al. 2006; Roth and Sullivan 2005). With the emergence of fear conditioning, this same procedure only produces learning-associated changes in the posterior piriform cortex, which has a well-documented role in learning in adults. While the role of the posterior piriform is not yet understood in pups, it does have highly distributed reciprocal interactions with the prefrontal, amygdaloid, entorhinal, and perirhinal cortices (Johnson et al. 2000), which suggests a major role in information synthesis (Wilson and Stevenson 2006; Haberly 2001).
15.3.5. Corticosterone (CORT) Increases Induce the Premature Emergence of Fear Conditioning
Perhaps the most convincing evidence that amygdala maturity does not account for pups emerging ability to learn fear is our ability to pharmacologically induce amygdala-dependent fear conditioning in pups as young as PN5 (Roth and Sullivan 2005; Moriceau and Sullivan 2004, 2006; Moriceau et al. 2006; Wiedenmayer and Barr 2001). Specifically, the-sensitive-period attachment learning required low corticosterone (CORT) during conditioning. Pups have a stress hyporesponsive period (SHRP) when stressors, such as shock, fail to induce a CORT increase (Grino et al. 1994; Levine 1962, 1967, 2001; Rosenfeld et al. 1992; Guillet and Michaelson 1978; Butte et al. 1973; Guillet et al. 1980; Cate and Yasumura 1975; Walker et al. 1986; Henning 1978). At around PN10, pups have a sufficient stress-induced CORT release to support fear conditioning (Moriceau et al. 2006). However, since maternal presence blocks shock-induced CORT release in these older pups, pups will continue to learn to prefer odors paired with shock if the mother is present during conditioning (Stanton et al. 1987; Stanton and Levine 1990; Suchecki et al. 1993; Moriceau and Sullivan 2006). In fact, these older pups can rapidly switch between the amygdala-dependent fear learning and attachment learning during odor-shock conditioning, dependent upon CORT levels modified by the mother (Moriceau and Sullivan 2006). While the mother’s ability to block stress-induced CORT release may appear unusual, it is fairly widespread in mammals. For example, maternal presence in adolescent guinea pigs, the presence of the mate in voles, the presence of peers in nonhuman primates, social affiliation in humans all reduce/block stress-induced CORT release (DeVries et al. 2003; Hennessy et al. 1995). With maturation, CORT takes on a more modulatory role of modifying the learned aversion and inhibitory conditioning (Corodimas et al. 1994; Hui et al. 2004; Pugh et al. 1997; Roozendaal et al. 1996, 2002; Thompson et al. 2004). Since early life stress prematurely increases pups CORT, early life stress prematurely terminates attachment learning and facilitates the precocious emergence of fear conditioning, which has recently been demonstrated in our laboratory (Moriceau et al. In preparation).
15.3.6. Development of Malaise Olfactory Learning
The developmental delay in pups’ emotional learning and amygdala-dependent fear conditioning is not due to pups’ inability to learn an odor avoidance. Indeed, young pups can learn to avoid odors, although malaise and introceptive distress rather than exteroceptive pain is responsible for this learning (Haroutunian and Campbell 1979). Specifically, rat pups can learn to avoid odors paired with malaise throughout ontogeny, including before birth (Shionoya et al. 2006; Smotherman 1982; Miller et al. 1990; Rudy and Cheatle 1983; Coopersmith and Leon 1986; Hoffmann et al. 1987). We have recently assessed the neurobiology of malaise learning. Since this learning occurs during the prenatal period, which is prior to the maturation of brain structures critical for adult odor-malaise learning (i.e., amygdala) (Touzani and Sclafani 2005; Berdel and Morys 2000; Berdel et al. 1997; Morys et al. 1998), we searched for a unique circuit for learning. We used two means of inducing malaise in pups, odor-LiCl and 1 mA high shock (Haroutunian and Campbell 1979), both of which also produce gastrointestinal distress in pups (Davenport 1950; Stern et al. 1969; Raineki et al. 2008). In sharp contrast to the important role of the amygdala in adult odor-malaise learning, young pups use the olfactory bulb and the piriform cortex to support learning (Shionoya et al. 2006). Indeed, it is not until pups are close to weaning that the amygdala is used in odor-malaise learning, which is almost a week later than the emergence of amygdala-dependent fear conditioning (Sullivan et al. 2000; Roth and Sullivan 2005; Moriceau and Sullivan 2006; Moriceau et al. 2006). Thus, despite the similar appearance in the odor-aversion learning produced by odor-0.5 mA fear conditioning and the odor-malaise learning, the neural circuitry supporting this learning differs, indicating caution is required when assuming that similar appearing behaviors in development use the same neural structures.
Maternal presence also modifies pups odor-malaise learning. Specifically, if pups are nursing while conditioned with odor-LiCl pairings, pups fail to learn the aversion and learn to prefer the odor (Shionoya et al. 2006). These data complement pups conditioning with taste-LiCl, which is blocked if pups are nursing (Martin and Alberts 1979; Gubernick and Alberts 1984; Melcer et al. 1985; Kehoe and Blass 1986).
As illustrated in Table 15.1, pups show remarkable odor learning abilities, which are uniquely adapted to the pups’ ecological demands as the transition from complete dependence changes to complete independence. Indeed, pups transition from unique neural circuits supporting their unique learning to circuits consistent with that documented in the adult. More remarkable is the dramatic effect of maternal presence on pup learning, which provides pups to “switch” to attachment learning in the mother’s presence. It is important to note that the similarity in behavior between fear and malaise learning (odor avoidance) suggests that similar behaviors across development may be an unreliable tool to assess brain development. Indeed, the age at which the amygdala supports pups’ emotional learning from odor-shock conditioning differs from the age the amygdala supports malaise learning, suggesting caution when extrapolating functional brain development from one behavior to another. Additionally, while this odor-0.5 mA shock conditioning begins to produce fear conditioning at PN10, fear conditioning is blocked and the early life odor preference learning is extended via suppression of the amygdala or if the mother is present (Moriceau and Sullivan 2006). A similar learning constraint exists with odor-LiCl and taste-LiCl learning. Specifically, if pups nurse during odor-LiCl conditioning, they develop a preference for the odor (Shionoya et al. 2006), while taste-LiCl avoidance learning is blocked (Martin and Alberts 1979; Gubernick and Alberts 1984; Melcer et al. 1985; Kehoe and Blass 1986).
TABLE 15.1
Infant Rats Show at Least Three Types of Learning in Early Life and Each Type of Learning Is Influenced by Maternal Presence, Albeit in Unique Ways.
15.4. IMPACT OF EARLY LIFE EVENTS ON ADULT ODOR MEMORY
Early life experiences have a profound effect on later life sensory, cognitive, and emotional processing. While a wide range of manipulations has been used to manipulate early life experiences (maternal deprivation, handling, maternal licking, odor-shock), there is remarkable convergence concerning mediating factors to induce enduring adult effects. Indeed, long-term effects have been documented in the hypothalamic-pituitary-adrenal (HPA) axis and the extended limbic system, including the LC, hippocampus, amygdala, and prefrontal cortex (Plotsky et al. 2005; Francis et al. 1999; Dent et al. 2001). While olfactory learning has not received much attention, adult cognitive function is well documented to be increased or decreased based on the type and age of infant manipulations (Fleming et al. 1999; Pryce et al. 2003; Denenberg and Bell 1960; Caldji et al. 1998, 2000; Coplan et al. 1996; Denenberg 1963, 1999; Kosten et al. 2005, 2006; Levine 1962, 1967, 2001; Lindholm 1962; Sevelinges et al. 2007, 2008; Seckl and Meaney 2004; Romeo et al. 2009).
We have been assessing the effects of infant odor learning on adult odor learning. However, as we assess the enduring effects of infant odor learning, it is important to remember that odor attachment learning differs from adult learning in that it involves social learning and attachment that occurs within the unique functioning of the infant brain. Specifically, the novel odor paired with either stroking or 0.5 mA shock takes on characteristics of maternal odor to support approach responses and complex motor responses such as nipple attachment. This odor produces a profound effect in infancy, which is retained into adulthood where the odor enhances male and female sexual behavior (Moore et al. 1996; Raineki et al. In progress; Coopersmith and Leon 1986; Fillion and Blass 1986; Sevelinges et al. 2007).
The attachment odor learned in infancy also modifies odor learning in adulthood (Sevelinges et al. 2007, 2008). Specifically, early life paired odor-shock conditioning attenuates adult fear conditioning and alters the functioning of olfactory sensory cortical areas, such as the piriform cortex and the basolateral amygdala. However, other changes were found even when the conditioned odor was not present. Specifically, using field potentials induced in the piriform cortex and the basolateral amygdala in response to paired-pulse stimulation of the olfactory bulb revealed that in these sites, the level of inhibition was significantly reduced in adult rats, but only if they had received infant paired odor-shock conditioning and not unpaired. These results suggest that the contingency or predictability of early life experience are important variables for an enduring effect of early life experience on adult cognition. Thus, infant experiences enduring effects are due, at least in part, to learning effects that interact with neural changes induced through experience to alter the trajectory of brain development.
15.5. CONCLUSION
This review of the neurobiology of emotional olfactory memory has focused on rat odor fear conditioning and highlights the fact that the olfactory modality might be particularly appropriate for understanding the interplay between limbic and cortical areas for the acquisition and storage of learned associations. Indeed, the existence of rapid direct anatomical connections between early sensory areas (i.e., the olfactory bulb and the piriform cortex) and the amygdala seems to favor the implication of olfactory cortical areas at early stages of learning acquisition. Together, these properties might endow the sense of smell with the capacity to automatically trigger emotional odor memories.
The developmental research reviewed here also illustrates the remarkable functional changes that occur in learning as an animal makes the transition from one developmental phase to the next. During early life, when infant rats are confined to the nest, they rapidly learn to prefer an odor when it is paired with myriad stimuli that appears dependent upon their ability to raise NE levels within the bulb rather than their hedonic value. This unique early life learning system may have evolved because of the critical survival value of odor learning: pups must learn the maternal odor to approach the mother and nipple attachment (Polan and Hofer 1999; Pedersen and Blass 1982; Leon 1992). Therefore, pups’ survival is dependent on odor approach learning, suggesting that evolutionary pressure may have developed a neural circuit to ensure pups only learn to approach their caregiver regardless of the quality of care received (Bowlby 1965; Hofer and Sullivan 2001). The development of odor learning provides insight into at least a few basic principles concerning the development of the neurobiology of learning: (1) learning that appears similar throughout development can be supported by neural systems showing very robust developmental changes; (2) the functional emergence of the amygdala, or perhaps any brain structure, during ontogeny depends upon which function of the brain area is being assessed; and (3) the context of learning (maternal presence) dramatically modifies the neural circuitry pups use for odor learning, and hence the behaviors learned.
The olfactory system is an evolutionarily old sensory system with a simplistic neural circuitry, at least compared to other sensory systems, that has direct access to the emotional centers of the brain. This suggests that the sense of smell has a critical role in controlling emotional behavior that has been maintained through evolution. Environmental odors control emotions and behavioral responses that enable avoidance of danger but approach in social situations, both of which enhance survival of the individual and the species. Importantly, the olfactory system is remarkably flexible and eloquently designed for emotional learning and memory, which is reflected in odors’ direct access to brain areas critical for emotional learning. Finally, the remarkable flexibility of the olfactory system to adapt to divergent learning demands at different developmental periods during the life span underscores the critical role of this sensory system in emotional and social attachment.
ACKNOWLEDGMENT
This work was funded by grants NICHD-HD33402, NSF IOB-0544406, NIDCD-CD009910, NIMH-MH086952, and Oklahoma Center for Science and Technology OCAST to RMS, and by grants ANR-05-PNRA-1.E7 AROMALIM to AMM.
REFERENCES
- Anagnostaras S.G., Gale G.D., Fanselow M.S. Hippocampus and contextual fear conditioning: Recent controversies and advances. Hippocampus. 2001;11(1):8–17. [PubMed: 11261775]
- Barnes D.C., Hofacer R.D., Zaman A.R., Rennaker R.L., Wilson D.A. Olfactory perceptual stability and discrimination. Nature Neuroscience. 2008;11(12):1378–80. [PMC free article: PMC2682180] [PubMed: 18978781]
- Barr G.A. Ontogeny of nociception and antinociception. NIDA Research Monogram. 1995;158:172–201. [PubMed: 8594484]
- Barr R., Marrott H., Rovee-Collier C. The role of sensory preconditioning in memory retrieval by preverbal infants. Learning & Behavior. 2003;31(2):111–23. [PubMed: 12882370]
- Berdel B., Morys J. Expression of calbindin-D28k and parvalbumin during development of rat’s basolateral amygdaloid complex. International Journal of Developmental Neuroscience. 2000;18(6):501–13. [PubMed: 10884595]
- Berdel B., Morys J., Maciejewska B. Neuronal changes in the basolateral complex during development of the amygdala of the rat. International Journal of Developmental Neuroscience. 1997;15(6):755–65. [PubMed: 9402226]
- Bernabeu R., Thiriet N., Zwiller J., Di Scala G. Lesion of the lateral entorhinal cortex amplifies odor-induced expression of c-fos, junB, and zif 268 mRNA in rat brain. Synapse. 2006;59(3):135–43. [PubMed: 16342059]
- Biella G., de Curtis M. Olfactory inputs activate the medial entorhinal cortex via the hippocampus. Journal of Neurophysiology. 2000;83(4):1924–31. [PubMed: 10758103]
- Biella G.R., Gnatkovsky V., Takashima I., Kajiwara R., Iijima T., de Curtis M. Olfactory input to the parahippocampal region of the isolated guinea pig brain reveals weak entorhinal-to-perirhinal interactions. European Journal of Neuroscience. 2003;18(1):95–101. [PubMed: 12859341]
- Blair H.T., Schafe G.E., Bauer E.P., Rodrigues S.M., LeDoux J.E. Synaptic plasticity in the lateral amygdala: A cellular hypothesis of fear conditioning. Learning & Memory. 2001;8(5):229–42. [PubMed: 11584069]
- Blozovski D., Cudennec A. Passive avoidance learning in the young rat. Developmental Psychobiology. 1980;13(5):513–18. [PubMed: 7409331]
- Boeijinga P.H., Van Groen T. Inputs from the olfactory bulb and olfactory cortex to the entorhinal cortex in the cat. II. Physiological studies. Experimental Brain Research. 1984;57(1):40–48. [PubMed: 6519229]
- Bouwmeester H., Wolterink G., van Ree J.M. Neonatal development of projections from the basolateral amygdala to prefrontal, striatal, and thalamic structures in the rat. Journal of Comparative Neurology. 2002;442(3):239–49. [PubMed: 11774339]
- Bowlby J. Attachment. New York: Basic Books; 1965.
- Brennan P.A. The nose knows who’s who: Chemosensory individuality and mate recognition in mice. Hormone and Behavior. 2004;46(3):231–40. [PubMed: 15325224]
- Brennan P.A., Kendrick K.M. Mammalian social odours: Attraction and individual recognition. Philosophical Transactions of the Royal Society London B Biological Sciences. 2006;361(1476):2061–78. [PMC free article: PMC1764843] [PubMed: 17118924]
- Brennan P.A., Keverne E.B. Neural mechanisms of mammalian olfactory learning. Progress in Neurobiology. 1997;51(4):457–81. [PubMed: 9106902]
- Brosh I., Rosenblum K., Barkai E. Learning-induced reversal of the effect of noradrenalin on the postburst AHP. Journal of Neurophysiology. 2006;96(4):1728–33. [PubMed: 16823026]
- Brothers L.A., Finch D.M. Physiological evidence for an excitatory pathway from entorhinal cortex to amygdala in the rat. Brain Research. 1985;359(1–2):10–20. [PubMed: 4075137]
- Brunjes P.C., Illig K.R., Meyer E.A. A field guide to the anterior olfactory nucleus (cortex). Brain Research Brain Research Review. 2005;50(2):305–35. [PubMed: 16229895]
- Buchel C., Dolan R.J. Classical fear conditioning in functional neuroimaging. Current Opinion in Neurobiology. 2000;10(2):219–23. [PubMed: 10753800]
- Butte J.C., Kakihana R., Farnham M.L., Noble E.P. The relationship between brain and plasma corticosterone stress response in developing rats. Endocrinology. 1973;92(6):1775–79. [PubMed: 4707271]
- Caldji C., Diorio J., Meaney M.J. Variations in maternal care in infancy regulate the development of stress reactivity. Biological Psychiatry. 2000;48(12):1164–74. [PubMed: 11137058]
- Caldji C., Tannenbaum B., Sharma S., Francis D., Plotsky P.M., Meaney M.J. Maternal care during infancy regulates the development of neural systems mediating the expression of fearfulness in the rat. Proceedings of the National Academy of Sciences of the United States of America. 1998;95(9):5335–40. [PMC free article: PMC20261] [PubMed: 9560276]
- Calu D.J., Roesch M.R., Stalnaker T.A., Schoenbaum G. Associative encoding in posterior piriform cortex during odor discrimination and reversal learning. Cerebral Cortex. 2007;17(6):1342–49. [PMC free article: PMC2473864] [PubMed: 16882682]
- Campbell B.A., Spear N.E. Ontogeny of memory. Psychological Review. 1972;79(3):215–36. [PubMed: 4341445]
- Carmichael S.T., Clugnet M.C., Price J.L. Central olfactory connections in the macaque monkey. Journal of Comparative Neurology. 1994;346(3):403–34. [PubMed: 7527806]
- Cate T.E., Yasumura S. Effects of ACTH and histamine stress on serum corticosterone and adrenal cyclic AMP in immature rats. Endocrinology. 1975;96:1044–1047. [PubMed: 164340]
- Chabaud P., Ravel N., Wilson D.A., Gervais R. Functional coupling in rat central olfactory pathways: A coherence analysis. Neuroscience Letters. 1999;276(1):17–20. [PubMed: 10586964]
- Chabaud P., Ravel N., Wilson D.A., Mouly A.M., Vigouroux M., Farget V., Gervais R. Exposure to behaviourally relevant odour reveals differential characteristics in rat central olfactory pathways as studied through oscillatory activities. Chemical Senses. 2000;25(5):561–73. [PubMed: 11015328]
- Cheng D.T., Knight D.C., Smith C.N., Stein E.A., Helmstetter F.J. Functional MRI of human amygdala activity during Pavlovian fear conditioning: Stimulus processing versus response expression. Behavioral Neuroscience. 2003;117(1):3–10. [PubMed: 12619902]
- Cheslock S.J., Varlinskaya E.I., Petrov E.S., Spear N.E. Rapid and robust olfactory conditioning with milk before suckling experience: Promotion of nipple attachment in the newborn rat. Behavioral Neuroscience. 2000;114(3):484–95. [PubMed: 10883799]
- Cheslock S.J., Varlinskaya E.I., High J.M., Spear N.E. Higher order conditioning in the newborn rat: Effects of temporal disparity imply infantile encoding of simultaneous events. Infancy. 2003;4(2):157–76.
- Chu S., Downes J.J. Odour-evoked autobiographical memories: Psychological investigations of proustian phenomena. Chemical Senses. 2000;25(1):111–16. [PubMed: 10668001]
- Chu S., Downes J.J. Proust nose best: Odors are better cues of autobiographical memory. Memory Cognition. 2002;30(4):511–18. [PubMed: 12184552]
- Collier A.C., Bolles R.C. The ontogenesis of defensive reactions to shock in preweanling rats. Developmental Psychobiology. 1980;13(2):141–50. [PubMed: 7188912]
- Collier A.C., Mast J. Alleviation of avoidance deficits by approach alternatives in 10-day old rats. Physiology & Behavior. 1979;23(3):615–18. [PubMed: 504456]
- Coopersmith R., Leon M. Enhanced neural response by adult rats to odors experienced early in life. Brain Research. 1986;371(2):400–3. [PubMed: 3697770]
- Coplan J.D., Andrews M.W., Rosenblum L.A., Owens M.J., Friedman S., Gorman J.M., Nemeroff C.B. Persistent elevations of cerebrospinal fluid concentrations of corticotropin-releasing factor in adult nonhuman primates exposed to early-life stressors: Implications for the pathophysiology of mood and anxiety disorders. Proceedings of the National Academy of Sciences of the United States of America. 1996;93(4):1619–23. [PMC free article: PMC39991] [PubMed: 8643680]
- Corodimas K.P., LeDoux J.E., Gold P.W., Schulkin J. Corticosterone potentiation of conditioned fear in rats. Annals of the New York Academy of Sciences. 1994;746:392–93. [PubMed: 7825895]
- Coureaud G., Moncomble A.S., Montigny D., Dewas M., Perrier G., Schaal B. A pheromone that rapidly promotes learning in the newborn. Current Opinion in Biology. 2006;16(19):1956–61. [PubMed: 17027493]
- Cousens G., Otto T. Both pre- and posttraining excitotoxic lesions of the basolateral amygdala abolish the expression of olfactory and contextual fear conditioning. Behavioral Neuroscience. 1998;112(5):1092–1103. [PubMed: 9829787]
- Datiche F., Cattarelli M. Reciprocal and topographic connections between the piriform and prefrontal cortices in the rat: A tracing study using the B subunit of the cholera toxin. Brain Research Bulletin. 1996;41(6):391–98. [PubMed: 8973845]
- Datiche F., Litaudon P., Cattarelli M. Intrinsic association fiber system of the piriform cortex: A quantitative study based on a cholera toxin B subunit tracing in the rat. Journal of Comparative Neurology. 1996;376(2):265–77. [PubMed: 8951642]
- Datiche F., Roullet F., Cattarelli M. Expression of Fos in the piriform cortex after acquisition of olfactory learning: An immunohistochemical study in the rat. Brain Research Bulletin. 2001;55(1):95–99. [PubMed: 11427343]
- Davenport V.D. Distribution of parenterally administered lithium in plasma, brain and muscle of rats. American Journal of Physiology. 1950;163(3):633–41. [PubMed: 14799641]
- Davis M., Walker D.L., Lee Y. Roles of the amygdala and bed nucleus of the stria terminalis in fear and anxiety measured with the acoustic startle reflex. Possible relevance to PTSD. Annals of the New York Academy of Sciences. 1997;821:305–31. [PubMed: 9238214]
- Davis M., Walker D.L., Myers K.M. Role of the amygdala in fear extinction measured with potentiated startle. Annals of the New York Academy of Sciences. 2003;985:218–32. [PubMed: 12724161]
- Davis M., Whalen P.J. The amygdala: Vigilance and emotion. Molecular Psychiatry. 2001;6(1):13–34. [PubMed: 11244481]
- de Olmos J., Hardy H., Heimer L. The afferent connections of the main and the accessory olfactory bulb formations in the rat: An experimental HRP-study. Journal of Comparative Neurology. 1978;181(2):213–44. [PubMed: 690266]
- Denenberg V.H. Early experience and emotional development. Scientific American. 1963;208:138–46. [PubMed: 14026976]
- Denenberg V.H. Commentary: Is maternal stimulation the mediator of the handling effect in infancy? Developmental Psychobiology. 1999;34(1):1–3. [PubMed: 9919428]
- Denenberg V.H., Bell R.W. Critical periods for the effects of infantile experience on adult learning. Science. 1960;131:227–28. [PubMed: 13815894]
- Dent G.W., Smith M.A., Levine S. Stress-induced alterations in locus coeruleus gene expression during ontogeny. Brain Research Developmental Brain Research. 2001;127(1):23–30. [PubMed: 11287061]
- DeVries A.C., Glasper E.R., Detillion C.E. Social modulation of stress responses. Physiology & Behavior. 2003;79(3):399–407. [PubMed: 12954434]
- Dielenberg R.A., McGregor I.S. Defensive behavior in rats toward predatory odors: A review. Neuroscience Biobehavioral Review. 2001;25(7–8):597–609. [PubMed: 11801285]
- Distel H., Hudson R. The contribution of the olfactory and tactile modalities to the nipple-search behaviour of newborn rabbits. Journal of Comparative Physiology. 1985;157(5):599–605. [PubMed: 3837101]
- Dziewiatkowski J., Spodnik J.H., Biranowska J., Kowianski P., Majak K., Morys J. The projection of the amygdaloid nuclei to various areas of the limbic cortex in the rat. Folia Morphology (Warsz). 1998;57(4):301–8. [PubMed: 10437306]
- Emerich D.F., Scalzo F.M., Enters E.K., Spear N.E., Spear L.P. Effects of 6-hydroxydopamine-induced catecholamine depletion on shock-precipitated wall climbing of infant rat pups. Developmental Psychobiology. 1985;18(3):215–27. [PubMed: 3921418]
- Ennis M., Linster C., Aroniadou-Anderjaska V., Ciombor K., Shipley M.T. Glutamate and synaptic plasticity at mammalian primary olfactory synapses. Annals of the New York Academy of Sciences. 1998;855:457–66. [PubMed: 9929639]
- Fanselow M.S., Gale G.D. The amygdala, fear, and memory. Annals of the New York Academy of Sciences. 2003;985:125–34. [PubMed: 12724154]
- Fanselow M.S., LeDoux J.E. Why we think plasticity underlying Pavlovian fear conditioning occurs in the basolateral amygdala. Neuron. 1999;23(2):229–32. [PubMed: 10399930]
- Ferry B., McGaugh J.L. Role of amygdala norepinephrine in mediating stress hormone regulation of memory storage. Acta Pharmacologica Sinica. 2000;21(6):481–93. [PubMed: 11360681]
- Fillion T.J., Blass E.M. Infantile experience with suckling odors determines adult sexual behavior in male rats. Science. 1986;231(4739):729–31. [PubMed: 3945807]
- Fitzgerald M. The development of nociceptive circuits. Nature Review Neuroscience. 2005;6(7):507–20. [PubMed: 15995722]
- Fleming A.S., O’Day D.H., Kraemer G.W. Neurobiology of mother-infant interactions: Experience and central nervous system plasticity across development and generations. Neuroscience Biobehavioral Review. 1999;23(5):673–85. [PubMed: 10392659]
- Fletcher M.L., Smith A.M., Best A.R., Wilson D.A. High-frequency oscillations are not necessary for simple olfactory discriminations in young rats. Journal of Neuroscience. 2005;25(4):792–98. [PMC free article: PMC2292205] [PubMed: 15673658]
- Francis D.D., Champagne F.A., Liu D., Meaney M.J. Maternal care, gene expression, and the development of individual differences in stress reactivity. Annals of the New York Academy of Sciences. 1999;896:66–84. [PubMed: 10681889]
- Funk D., Amir S. Enhanced fos expression within the primary olfactory and limbic pathways induced by an aversive conditioned odor stimulus. Neuroscience. 2000;98(3):403–6. [PubMed: 10869835]
- Gilles E.E., Schultz L., Baram T.Z. Abnormal corticosterone regulation in an immature rat model of continuous chronic stress. Pediatric Neurology. 1996;15(2):114–19. [PMC free article: PMC3415889] [PubMed: 8888044]
- Gottfried J.A., Winston J.S., Dolan R.J. Dissociable codes of odor quality and odorant structure in human piriform cortex. Neuron. 2006;49(3):467–79. [PubMed: 16446149]
- Grino M., Paulmyer-Lacroix O., Faudon M., Renard M., Anglade G. Blockade of alpha 2-adrenoceptors stimulates basal and stress-induced adrenocorticotropin secretion in the developing rat through a central mechanism independent from corticotropin-releasing factor and arginine vasopressin. Endocrinology. 1994;135(6):2549–57. [PubMed: 7988443]
- Gubernick D.J., Alberts J.R. A specialization of taste aversion learning during suckling and its weaning-associated transformation. Developmental Psychobiology. 1984;17(6):613–28. [PubMed: 6096189]
- Guillet R., Michaelson S.M. Corticotropin responsiveness in the neonatal rat. Neuroendocrinology. 1978;27(3–4):119–25. [PubMed: 215931]
- Guillet R., Saffran M., Michaelson S.M. Pituitary-adrenal response in neonatal rats. Endocrinology. 1980;106(3):991–94. [PubMed: 6243561]
- Guthrie K.M., Gall C. Anatomic mapping of neuronal odor responses in the developing rat olfactory bulb. Journal of Comparative Neurology. 2003;455(1):56–71. [PubMed: 12454996]
- Guthrie K.M., Gall C.M. Functional mapping of odor-activated neurons in the olfactory bulb. Chemical Senses. 1995;20(2):271–82. [PubMed: 7583022]
- Haberly L.B. Parallel-distributed processing in olfactory cortex: New insights from morphological and physiological analysis of neuronal circuitry. Chemical Senses. 2001;26(5):551–76. [PubMed: 11418502]
- Haberly L.B., Price J.L. The axonal projection patterns of the mitral and tufted cells of the olfactory bulb in the rat. Brain Research. 1977;129(1):152–57. [PubMed: 68803]
- Haberly L.B., Price J.L. Association and commissural fiber systems of the olfactory cortex of the rat. Journal of Comparative Neurology. 1978;178(4):711–40. [PubMed: 632378]
- Hall J., Thomas K.L., Everitt B.J. Rapid and selective induction of BDNF expression in the hippocampus during contextual learning. Nature Neuroscience. 2000;3(6):533–35. [PubMed: 10816306]
- Harley C.W., Darby-King A., McCann J., McLean J.H. Beta1-adrenoceptor or alpha1-adrenoceptor activation initiates early odor preference learning in rat pups: Support for the mitral cell/cAMP model of odor preference learning. Learning & Memory. 2006;13(1):8–13. [PubMed: 16452650]
- Harlow H.F., Harlow M.K. The affectional systems. In: Schrier A., Harlow H.F., Stollnitz F., editors. Behavior of Nonhuman Primates. New York: Academic Press; 1965. pp. 287–355.
- Haroutunian V., Campbell B.A. Emergence of interoceptive and exteroceptive control of behavior in rats. Science. 1979;205(4409):927–29. [PubMed: 472715]
- Hégoburu C., Sevelinges Y., Thévenet M., Gervais R., Parrot S., Mouly A.M. Dynamics of glutamate and GABA variations in amygdala and olfactory cortex inside odore fear acquisition session in rats. Submitted. [PubMed: 19864295]
- Helfer M.E., Kempe R.S., Krugman R.D. The Battered Child. Chicago, IL: University of Chicago Press; 1997.
- Hennessy M.B., Nigh C.K., Sims M.L., Long S.J. Plasma cortisol and vocalization responses of postweaning age guinea pigs to maternal and sibling separation: Evidence for filial attachment after weaning. Developmental Psychobiology. 1995;28(2):103–15. [PubMed: 8529784]
- Henning S.J. Plasma concentrations of total and free corticosterone during development in the rat. American Journal of Physiology. 1978;235(5):E451–56. [PubMed: 103438]
- Herz R.S. Are odors the best cues to memory? A cross-modal comparison of associative memory stimuli. Annals of the New York Academy of Sciences. 1998;855:670–74. [PubMed: 9929669]
- Herz R.S., Cupchik G.C. The emotional distinctiveness of odor-evoked memories. Chemical Senses. 1995;20(5):517–28. [PubMed: 8564426]
- Herz R.S., Schooler J.W. A naturalistic study of autobiographical memories evoked by olfactory and visual cues: Testing the Proustian hypothesis. American Journal of Psychology. 2002;115(1):21–32. [PubMed: 11868193]
- Herzog C., Otto T. Odor-guided fear conditioning in rats: 2. Lesions of the anterior perirhinal cortex disrupt fear conditioned to the explicit conditioned stimulus but not to the training context. Behavioral Neuroscience. 1997;111(6):1265–72. [PubMed: 9438795]
- Herzog C., Otto T. Contributions of anterior perirhinal cortex to olfactory and contextual fear conditioning. Neuroreport. 1998;9(8):1855–59. [PubMed: 9665615]
- Hofer M.A., Shair H., Singh P. Evidence that maternal ventral skin substances promote suckling in infant rats. Physiology & Behavior. 1976;17(1):131–36. [PubMed: 1033575]
- Hofer M.A., Sullivan R.M. Toward a neurobiology of attachment. In: Nelson C.A., Luciana M., editors. Developmental Cognitive Neuroscience. Cambridge, MA: MIT Press; 2001. pp. 599–616.
- Hofer M.A., Sullivan R.M. Toward a neurobiology of attachment. In: Nelson C.A., Luciana M., editors. Developmental Cognitive Neuroscience. Cambridge, MA: MIT Press; 2008.
- Hoffmann H., Molina J.C., Kucharski D., Spear N.E. Further examination of ontogenetic limitations on conditioned taste aversion. Developmental Psychobiology. 1987;20(4):455–63. [PubMed: 3038640]
- Hoffmann H., Spear N.E. Ontogenetic differences in conditioning of an aversion to a gustatory CS with a peripheral US. Behavioral and Neural Biology. 1988;50(1):16–23. [PubMed: 3401194]
- Hui G.K., Figueroa I.R., Poytress B.S., Roozendaal B., McGaugh J.L., Weinberger N.M. Memory enhancement of classical fear conditioning by post-training injections of corticosterone in rats. Neurobiology of Learning & Memory. 2004;81(1):67–74. [PubMed: 14670360]
- Hunt P.S., Richardson R., Campbell B.A. Delayed development of fear-potentiated startle in rats. Behavioral Neuroscience. 1994;108(1):69–80. [PubMed: 8192852]
- Illig K.R. Projections from orbitofrontal cortex to anterior piriform cortex in the rat suggest a role in olfactory information processing. Journal of Comparative Neurology. 2005;488(2):224–31. [PMC free article: PMC1360190] [PubMed: 15924345]
- Insausti R., Herrero M.T., Witter M.P. Entorhinal cortex of the rat: Cytoarchitectonic subdivisions and the origin and distribution of cortical efferents. Hippocampus. 1997;7(2):146–83. [PubMed: 9136047]
- Johnson B.A., Woo C.C., Duong H., Nguyen V., Leon M. A learned odor evokes an enhanced Fos-like glomerular response in the olfactory bulb of young rats. Brain Research. 1995;699(2):192–200. [PubMed: 8616621]
- Johnson D.M., Illig K.R., Behan M., Haberly L.B. New features of connectivity in piriform cortex visualized by intracellular injection of pyramidal cells suggest that ‘primary’ olfactory cortex functions like ‘association’ cortex in other sensory systems. Journal of Neuroscience. 2000;20(18):6974–82. [PMC free article: PMC6772836] [PubMed: 10995842]
- Jones S.V., Stanek-Rattiner L., Davis M., Ressler K.J. Differential regional expression of brain-derived neurotrophic factor following olfactory fear learning. Learning & Memory. 2007;14(12):816–20. [PMC free article: PMC2444033] [PubMed: 18086824]
- Jung M.W., Larson J., Lynch G. Long-term potentiation of monosynaptic EPSPs in rat piriform cortex in vitro. Synapse. 1990;6(3):279–83. [PubMed: 1978422]
- Kadohisa M., Wilson D.A. Olfactory cortical adaptation facilitates detection of odors against background. Journal of Neurophysiology. 2006;95(3):1888–96. [PMC free article: PMC2292127] [PubMed: 16251260]
- Kadohisa M., Wilson D.A. Separate encoding of identity and similarity of complex familiar odors in piriform cortex. Proceedings of the National Academy of Sciences of the United States of America. 2006;103(41):15206–11. [PMC free article: PMC1622801] [PubMed: 17005727]
- Kehoe P., Blass E.M. Conditioned aversions and their memories in 5-day-old rats during suckling. Journal of Experimental Psychology Animal Behavior Process. 1986;12(1):40–47. [PubMed: 3009681]
- Keller M., Meurisse M., Levy F. Mapping the neural substrates involved in maternal responsiveness and lamb olfactory memory in parturient ewes using Fos imaging. Behavioral Neuroscience. 2004;118(6):1274–84. [PubMed: 15598136]
- Keller M., Meurisse M., Levy F. Mapping of brain networks involved in consolidation of lamb recognition memory. Neuroscience. 2005;133(2):359–69. [PubMed: 15885919]
- Kesslak J.P., So V., Choi J., Cotman C.W., Gomez-Pinilla F. Learning upregulates brain-derived neurotrophic factor messenger ribonucleic acid: A mechanism to facilitate encoding and circuit maintenance? Behavioral Neuroscience. 1998;112(4):1012–19. [PubMed: 9733207]
- Kilpatrick L., Cahill L. Modulation of memory consolidation for olfactory learning by reversible inactivation of the basolateral amygdala. Behavioral Neuroscience. 2003;117(1):184–88. [PubMed: 12619920]
- Kolb B., Petrie B., Cioe J. Recovery from early cortical damage in rats, VII. Comparison of the behavioural and anatomical effects of medial prefrontal lesions at different ages of neural maturation. Behavior Brain Research. 1996;79(1–2):1–14. [PubMed: 8883811]
- Kosten T.A., Lee H.J., Kim J.J. Early life stress impairs fear conditioning in adult male and female rats. Brain Research. 2006;1087(1):142–50. [PubMed: 16626646]
- Kosten T.A., Miserendino M.J., Bombace J.C., Lee H.J., Kim J.J. Sex-selective effects of neonatal isolation on fear conditioning and foot shock sensitivity. Behavior Brain Research. 2005;157(2):235–44. [PubMed: 15639174]
- Krettek J.E., Price J.L. Amygdaloid projections to subcortical structures within the basal forebrain and brainstem in the rat and cat. Journal of Comparative Neurology. 1978;178(2):225–54. [PubMed: 627625]
- LaBar K.S., Gatenby J.C., Gore J.C., LeDoux J.E., Phelps E.A. Human amygdala activation during conditioned fear acquisition and extinction: A mixed-trial fMRI study. Neuron. 1998;20(5):937–45. [PubMed: 9620698]
- Laviolette S.R., Lipski W.J., Grace A.A. A subpopulation of neurons in the medial prefrontal cortex encodes emotional learning with burst and frequency codes through a dopamine D4 receptor-dependent basolateral amygdala input. Journal of Neuroscience. 2005;25(26):6066–75. [PMC free article: PMC6725053] [PubMed: 15987936]
- Le Jeune H., Jourdan F. Postnatal development of cholinergic markers in the rat olfactory bulb: A histochemical and immunocytochemical study. Journal of Comparative Neurology. 1991;314(2):383–95. [PubMed: 1787181]
- LeDoux J.E. Emotion circuits in the brain. Annual Review of Neuroscience. 2000;23:155–84. [PubMed: 10845062]
- Leon M. The neurobiology of filial learning. Annual Review of Psychology. 1992;43:377–98. [PubMed: 1539947]
- Leon M., Johnson B.A. Olfactory coding in the mammalian olfactory bulb. Brain Research Brain Research Review. 2003;42(1):23–32. [PubMed: 12668289]
- Levine S. Plasma-free corticosteroid response to electric shock in rats stimulated in infancy. Science. 1962;135:795–96. [PubMed: 14464660]
- Levine S. Maternal and environmental influences on the adrenocortical response to stress in weanling rats. Science. 1967;156(772):258–60. [PubMed: 6021047]
- Levine S. Primary social relationships influence the development of the hypothalamic–pituitary–adrenal axis in the rat. Physiology & Behavior. 2001;73(3):255–60. [PubMed: 11438350]
- Levy F., Keller M., Poindron P. Olfactory regulation of maternal behavior in mammals. Hormone and Behavior. 2004;46(3):284–302. [PubMed: 15325229]
- Li W., Howard J.D., Parrish T.B., Gottfried J.A. Aversive learning enhances perceptual and cortical discrimination of indiscriminable odor cues. Science. 2008;319(5871):1842–45. [PMC free article: PMC2756335] [PubMed: 18369149]
- Li W., Luxenberg E., Parrish T., Gottfried J.A. Learning to smell the roses: Experience-dependent neural plasticity in human piriform and orbitofrontal cortices. Neuron. 2006;52(6):1097–1108. [PMC free article: PMC1779760] [PubMed: 17178411]
- Lindholm B.W. Critical periods and the effects of early shock on later emotional behavior in the white rat. Journal of Comparative Physiology & Psychology. 1962;55:597–99. [PubMed: 14465479]
- Litaudon P., Amat C., Bertrand B., Vigouroux M., Buonviso N. Piriform cortex functional heterogeneity revealed by cellular responses to odours. European Journal of Neuroscience. 2003;17(11):2457–61. [PubMed: 12814377]
- Litaudon P., Cattarelli M. Piriform cortex late activity revealed functional spatial heterogeneity. Neuroreport. 1995;6(10):1377–80. [PubMed: 7488728]
- Litaudon P., Cattarelli M. Olfactory bulb repetitive stimulations reveal non-homogeneous distribution of the inhibitory processes in the rat piriform cortex. European Journal of Neuroscience. 1996;8(1):21–29. [PubMed: 8713447]
- Lopez-Fernandez M.A., Montaron M.F., Varea E., Rougon G., Venero C., Abrous D.N., Sandi C. Upregulation of polysialylated neural cell adhesion molecule in the dorsal hippocampus after contextual fear conditioning is involved in long-term memory formation. Journal of Neuroscience. 2007;27(17):4552–61. [PMC free article: PMC6673006] [PubMed: 17460068]
- Luskin M.B., Price J.L. The topographic organization of associational fibers of the olfactory system in the rat, including centrifugal fibers to the olfactory bulb. Journal of Comparative Neurology. 1983;216(3):264–91. [PubMed: 6306065]
- Maestripieri D., Tomaszycki M., Carroll K.A. Consistency and change in the behavior of rhesus macaque abusive mothers with successive infants. Developmental Psychobiology. 1999;34(1):29–35. [PubMed: 9919431]
- Majak K., Ronkko S., Kemppainen S., Pitkanen A. Projections from the amygdaloid complex to the piriform cortex: A PHA-L study in the rat. Journal of Comparative Neurology. 2004;476(4):414–28. [PubMed: 15282713]
- Maren S. Neurobiology of Pavlovian fear conditioning. Annual Review of Neuroscience. 2001;24:897–931. [PubMed: 11520922]
- Maren S. The amygdala, synaptic plasticity, and fear memory. Annals of the New York Academy of Sciences. 2003;985:106–13. [PubMed: 12724152]
- Maren S., Aharonov G., Fanselow M.S. Neurotoxic lesions of the dorsal hippocampus and Pavlovian fear conditioning in rats. Behavior Brain Research. 1997;88(2):261–74. [PubMed: 9404635]
- Maren S., Quirk G.J. Neuronal signalling of fear memory. Nature Review Neuroscience. 2004;5(11):844–52. [PubMed: 15496862]
- Martin C., Gervais R., Chabaud P., Messaoudi B., Ravel N. Learning-induced modulation of oscillatory activities in the mammalian olfactory system: The role of the centrifugal fibres. Journal of Physiology Paris. 2004;98(4–6):467–78. [PubMed: 16274975]
- Martin C., Gervais R., Messaoudi B., Ravel N. Learning-induced oscillatory activities correlated to odour recognition: A network activity. European Journal of Neuroscience. 2006;23(7):1801–10. [PubMed: 16623837]
- Martin L.T., Alberts J.R. Taste aversions to mother’s milk: The age-related role of nursing in acquisition and expression of a learned association. Journal of Comparative Physiology & Psychology. 1979;93(3):430–45. [PubMed: 479392]
- McDonald A.J. Cortical pathways to the mammalian amygdala. Progress in Neurobiology. 1998;55(3):257–332. [PubMed: 9643556]
- McDonald A.J., Mascagni F. Projections of the lateral entorhinal cortex to the amygdala: A Phaseolus vulgaris leucoagglutinin study in the rat. Neuroscience. 1997;77(2):445–59. [PubMed: 9472403]
- McGaugh J.L. Make mild moments memorable: Add a little arousal. Trends Cognition Science. 2006;10(8):345–47. [PubMed: 16793325]
- McLean J.H., Shipley M.T. Postnatal development of the noradrenergic projection from locus coeruleus to the olfactory bulb in the rat. Journal of Comparative Neurology. 1991;304(3):467–77. [PubMed: 2022760]
- Melcer T., Alberts J.R., Gubernick D.J. Early weaning does not accelerate the expression of nursing-related taste aversions. Developmental Psychobiology. 1985;18(5):375–81. [PubMed: 4065427]
- Milad M.R., Quirk G.J. Neurons in medial prefrontal cortex signal memory for fear extinction. Nature. 2002;420(6911):70–74. [PubMed: 12422216]
- Miller J.S., Molina J.C., Spear N.E. Ontogenetic differences in the expression of odor-aversion learning in 4- and 8-day-old rats. Developmental Psychobiology. 1990;23(4):319–30. [PubMed: 2170214]
- Moore C.L., Jordan L., Wong L. Early olfactory experience, novelty, and choice of sexual partner by male rats. Physiology & Behavior. 1996;60(5):1361–67. [PubMed: 8916195]
- Moriceau S., Sullivan R.M. Lack of CRF in to the locus coeruleus may underline neonatal rats failure to learn an odor avoidance. International Society for Development Psychobiology Annual Meeting, Orlando. 2002
- Moriceau S., Sullivan R.M. Corticosterone influences on Mammalian neonatal sensitive-period learning. Behavioral Neuroscience. 2004;118(2):274–81. [PMC free article: PMC1868531] [PubMed: 15113251]
- Moriceau S., Sullivan R.M. Unique neural circuitry for neonatal olfactory learning. Journal of Neuroscience. 2004;24(5):1182–89. [PMC free article: PMC1868533] [PubMed: 14762136]
- Moriceau S., Sullivan R.M. Maternal presence serves as a switch between learning fear and attraction in infancy. Nature Neuroscience. 2006;9(8):1004–6. [PMC free article: PMC1560090] [PubMed: 16829957]
- Moriceau S., Wilson D.A., Levine S., Sullivan R.M. Dual circuitry for odor-shock conditioning during infancy: Corticosterone switches between fear and attraction via amygdala. Journal of Neuroscience. 2006;26(25):6737–48. [PMC free article: PMC1574366] [PubMed: 16793881]
- Morys J., Berdel B., Jagalska-Majewska H., Luczynska A. The basolateral amygdaloid complex—its development, morphology and functions. Folia Morphology (Warsz). 1999;58(3 Suppl 2):29–46. [PubMed: 10959259]
- Morys J., Berdel B., Kowianski P., Dziewiatkowski J. The pattern of synaptophysin changes during the maturation of the amygdaloid body and hippocampal hilus in the rat. Folia Neuropathology. 1998;36(1):15–23. [PubMed: 9595859]
- Mouly A.M., Di Scala G. Entorhinal cortex stimulation modulates amygdala and piriform cortex responses to olfactory bulb inputs in the rat. Neuroscience. 2006;137(4):1131–41. [PubMed: 16325349]
- Mouly A.M., Fort A., Ben-Boutayab N., Gervais R. Olfactory learning induces differential long-lasting changes in rat central olfactory pathways. Neuroscience. 2001;102(1):11–21. [PubMed: 11226666]
- Mouly A.M., Gervais R. Polysynaptic potentiation at different levels of rat olfactory pathways following learning. Learning & Memory. 2002;9(2):66–75. [PMC free article: PMC155934] [PubMed: 11992017]
- Mouly A.M., Litaudon P., Chabaud P., Ravel N., Gervais R. Spatiotemporal distribution of a late synchronized activity in olfactory pathways following stimulation of the olfactory bulb in rats. European Journal of Neuroscience. 1998;10(3):1128–35. [PubMed: 9753181]
- Myers K.M., Davis M. Mechanisms of fear extinction. Molecular Psychiatry. 2007;12(2):120–50. [PubMed: 17160066]
- Nakamura S., Sakaguchi T. Development and plasticity of the locus coeruleus: A review of recent physiological and pharmacological experimentation. Progress in Neurobiology. 1990;34(6):505–26. [PubMed: 2202018]
- Nakamura S., Sakaguchi T., Kimura F., Aoki F. The role of alpha 1-adrenoceptor-mediated collateral excitation in the regulation of the electrical activity of locus coeruleus neurons. Neuroscience. 1988;27(3):921–29. [PubMed: 2855264]
- Nemeroff C.B. Neurobiological consequences of childhood trauma. Journal of Clinical Psychiatry. 2004;65(1):18–28. [PubMed: 14728093]
- Nowak R., Keller M., Val-Laillet D., Levy F. Perinatal visceral events and brain mechanisms involved in the development of mother-young bonding in sheep. Hormone and Behavior. 2007;52(1):92–98. [PubMed: 17488646]
- Ottersen O.P. Connections of the amygdala of the rat. IV: Corticoamygdaloid and intraamygdaloid connections as studied with axonal transport of horseradish peroxidase. Journal of Comparative Neurology. 1982;205(1):30–48. [PubMed: 7068948]
- Otto T., Cousens G., Herzog C. Behavioral and neuropsychological foundations of olfactory fear conditioning. Behavior Brain Research. 2000;110(1–2):119–28. [PubMed: 10802309]
- Otto T., Cousens G., Rajewski K. Odor-guided fear conditioning in rats: 1. Acquisition, retention, and latent inhibition. Behavioral Neuroscience. 1997;111(6):1257–64. [PubMed: 9438794]
- Otto T., Poon P. Dorsal hippocampal contributions to unimodal contextual conditioning. Journal of Neuroscience. 2006;26(24):6603–9. [PMC free article: PMC6674031] [PubMed: 16775148]
- Pape H.C., Stork O. Genes and mechanisms in the amygdala involved in the formation of fear memory. Annals of the New York Academy of Sciences. 2003;985:92–105. [PubMed: 12724151]
- Pare D., Quirk G.J., Ledoux J.E. New vistas on amygdala networks in conditioned fear. Journal of Neurophysiology. 2004;92(1):1–9. [PubMed: 15212433]
- Parsons T.C., Otto T. Temporary inactivation of dorsal hippocampus attenuates explicitly nonspatial, unimodal, contextual fear conditioning. Neurobiology of Learning & Memory. 2008;90(1):261–68. [PubMed: 18485754]
- Paschall G.Y., Davis M. Olfactory-mediated fear-potentiated startle. Behavioral Neuroscience. 2002;116(1):4–12. [PubMed: 11895182]
- Pedersen P.E., Blass E.M. Prenatal and postnatal determinants of the 1st suckling episode in albino rats. Developmental Psychobiology. 1982;15(4):349–55. [PubMed: 7106394]
- Pedersen P.E., Williams C.L., Blass E.M. Activation and odor conditioning of suckling behavior in 3-day-old albino rats. Journal of Experimental Psychology Animal Behavior Process. 1982;8(4):329–41. [PubMed: 7175445]
- Phillips R.G., LeDoux J.E. Differential contribution of amygdala and hippocampus to cued and contextual fear conditioning. Behavioral Neuroscience. 1992;106(2):274–85. [PubMed: 1590953]
- Plailly J., Howard J.D., Gitelman D.R., Gottfried J.A. Attention to odor modulates thalamocortical connectivity in the human brain. Journal of Neuroscience. 2008;28(20):5257–67. [PMC free article: PMC2706104] [PubMed: 18480282]
- Plotsky P.M., Thrivikraman K.V., Nemeroff C.B., Caldji C., Sharma S., Meaney M.J. Long-term consequences of neonatal rearing on central corticotropin-releasing factor systems in adult male rat offspring. Neuropsychopharmacology. 2005;30(12):2192–2204. [PubMed: 15920504]
- Polan H.J., Hofer M.A. Maternally directed orienting behaviors of newborn rats. Developmental Psychobiology. 1999;34(4):269–79. [PubMed: 10331151]
- Price J.L. An autoradiographic study of complementary laminar patterns of termination of afferent fibers to the olfactory cortex. Journal of Comparative Neurology. 1973;150(1):87–108. [PubMed: 4722147]
- Price J.L., Slotnick B.M., Revial M.F. Olfactory projections to the hypothalamus. Journal of Comparative Neurology. 1991;306(3):447–61. [PubMed: 1713925]
- Proust M. Swann’s way in. Paris: Grasset; 1913. In search of Lost Time.
- Pryce C.R., Bettschen D., Nanz-Bahr N.I., Feldon J. Comparison of the effects of early handling and early deprivation on conditioned stimulus, context, and spatial learning and memory in adult rats. Behavioral Neuroscience. 2003;117(5):883–93. [PubMed: 14570539]
- Pugh C.R., Tremblay D., Fleshner M., Rudy J.W. A selective role for corticosterone in contextual-fear conditioning. Behavioral Neuroscience. 1997;111(3):503–11. [PubMed: 9189265]
- Raineki C., Moriceau S., Boggs J., Sullivan R.M. Developing a neurobehavioral animal model infant’s attachment of an abusive caregiver. [PMC free article: PMC3929962] [PubMed: 20163787]
- Raineki C., Shionoya K., Sander K., Sullivan R.M. Ontogeny of odor-LiCl vs odor-shock learning: Similar behaviors but divergent ages of functional amygdala emergence. Learning & Memory. 2009;16(2):114–21. [PMC free article: PMC2661245] [PubMed: 19181617]
- Rangel S., Leon M. Early odor preference training increases olfactory bulb norepinephrine. Brain Research Developmental Brain Research. 1995;85(2):187–91. [PubMed: 7600666]
- Rattiner L.M., Davis M., French C.T., Ressler K.J. Brain-derived neurotrophic factor and tyrosine kinase receptor B involvement in amygdala-dependent fear conditioning. Journal of Neuroscience. 2004;24(20):4796–4806. [PMC free article: PMC6729469] [PubMed: 15152040]
- Ray J.P., Price J.L. The organization of the thalamocortical connections of the mediodorsal thalamic nucleus in the rat, related to the ventral forebrain-prefrontal cortex topography. Journal of Comparative Neurology. 1992;323(2):167–97. [PubMed: 1401255]
- Rescorla R.A. Pavlovian conditioning and its proper control procedures. Psychological Review. 1967;74(1):71–80. [PubMed: 5341445]
- Rescorla R.A. Behavioral studies of Pavlovian conditioning. Annual Review of Neuroscience. 1988;11:329–52. [PubMed: 3284445]
- Ressler K.J., Paschall G., Zhou X.L., Davis M. Regulation of synaptic plasticity genes during consolidation of fear conditioning. Journal of Neuroscience. 2002;22(18):7892–7902. [PMC free article: PMC6758105] [PubMed: 12223542]
- Richardson R., Tronson N., Bailey G.K., Parnas A.S. Extinction of conditioned odor potentiation of startle. Neurobiology of Learning & Memory. 2002;78(2):426–40. [PubMed: 12431427]
- Richardson R., Vishney A., Lee J. Conditioned odor potentiation of startle in rats. Behavioral Neuroscience. 1999;113(4):787–94. [PubMed: 10495086]
- Roesch M.R., Stalnaker T.A., Schoenbaum G. Associative encoding in anterior piriform cortex versus orbitofrontal cortex during odor discrimination and reversal learning. Cerebral Cortex. 2007;17(3):643–52. [PMC free article: PMC2396586] [PubMed: 16699083]
- Romeo R.D., Tang A.C., Sullivan R.M. Early life experiences: Enduring behavioral and endocrinological consequences. In: Pfaff D., Arnold A., editors. Hormones, Brain and Behavior. San Diego, CA: Elsevier; 2009.
- Roozendaal B., Brunson K.L., Holloway B.L., McGaugh J.L., Baram T.Z. Involvement of stress-released corticotropin-releasing hormone in the basolateral amygdala in regulating memory consolidation. Proceedings of the National Academy of Sciences of the United States of America. 2002;99(21):13908–13. [PMC free article: PMC129796] [PubMed: 12361983]
- Roozendaal B., Carmi O., McGaugh J.L. Adrenocortical suppression blocks the memory-enhancing effects of amphetamine and epinephrine. Proceedings of the National Academy of Sciences of the United States of America. 1996;93(4):1429–33. [PMC free article: PMC39955] [PubMed: 8643648]
- Rosen J.B., Schulkin J. From normal fear to pathological anxiety. Psychological Review. 1998;105(2):325–50. [PubMed: 9577241]
- Rosenfeld P., Suchecki D., Levine S. Multifactorial regulation of the hypothalamic-pituitary-adrenal axis during development. Neuroscience Biobehavioral Review. 1992;16(4):553–68. [PubMed: 1480351]
- Rosenkranz J.A., Grace A.A. Dopamine-mediated modulation of odour-evoked amygdala potentials during pavlovian conditioning. Nature. 2002;417(6886):282–87. [PubMed: 12015602]
- Roth T.L., Sullivan R.M. Memory of early maltreatment: Neonatal behavioral and neural correlates of maternal maltreatment within the context of classical conditioning. Biological Psychiatry. 2005;57(8):823–31. [PubMed: 15820702]
- Rudy J.W., Cheatle M.D. Odor-aversion learning by rats following LiCl exposure: Ontogenetic influences. Developmental Psychobiology. 1983;16(1):13–22. [PubMed: 6298037]
- Rush A.N., Robinette B.L., Stanton M.E. Ontogenetic differences in the effects of unpaired stimulus preexposure on eyeblink conditioning in the rat. Developmental Psychobiology. 2001;39(1):8–18. [PubMed: 11507705]
- Salzen E.A. Imprinting in birds and primates. Behaviour. 1967;28:232–254. [PubMed: 4962581]
- Sananes C.B., Campbell B.A. Role of the central nucleus of the amygdala in olfactory heart rate conditioning. Behavioral Neuroscience. 1989;103(3):519–25. [PubMed: 2736066]
- Sanchez M.M., Ladd C.O., Plotsky P.M. Early adverse experience as a developmental risk factor for later psychopathology: Evidence from rodent and primate models. Development & Psychopathology. 2001;13(3):419–49. [PubMed: 11523842]
- Sara S.J., Vankov A., Herve A. Locus coeruleus-evoked responses in behaving rats: A clue to the role of noradrenaline in memory. Brain Research Bulletin. 1994;35(5–6):457–65. [PubMed: 7859103]
- Savander V., LeDoux J.E., Pitkanen A. Topographic projections from the periamygdaloid cortex to select subregions of the lateral nucleus of the amygdala in the rat. Neuroscience Letters. 1996;211(3):167–70. [PubMed: 8817567]
- Schaal B., Coureaud G., Langlois D., Ginies C., Semon E., Perrier G. Chemical and behavioural characterization of the rabbit mammary pheromone. Nature. 2003;424(6944):68–72. [PubMed: 12840760]
- Schaal B., Marlier L., Soussignan R. Human foetuses learn odours from their pregnant mother’s diet. Chemical Senses. 2000;25(6):729–37. [PubMed: 11114151]
- Schafe G.E., Nader K., Blair H.T., LeDoux J.E. Memory consolidation of Pavlovian fear conditioning: A cellular and molecular perspective. Trends Neuroscience. 2001;24(9):540–46. [PubMed: 11506888]
- Schettino L.F., Otto T. Patterns of Fos expression in the amygdala and ventral perirhinal cortex induced by training in an olfactory fear conditioning paradigm. Behavioral Neuroscience. 2001;115(6):1257–72. [PubMed: 11770057]
- Schwob J.E., Price J.L. The development of lamination of afferent fibers to the olfactory cortex in rats, with additional observations in the adult. Journal of Comparative Neurology. 1984;223(2):203–22. [PubMed: 6200519]
- Seckl J.R., Meaney M.J. Glucocorticoid programming. Annals of the New York Academy of Sciences. 2004;1032:63–84. [PubMed: 15677396]
- Sevelinges Y., Gervais R., Messaoudi B., Granjon L., Mouly A.M. Olfactory fear conditioning induces field potential potentiation in rat olfactory cortex and amygdala. Learning & Memory. 2004;11(6):761–69. [PMC free article: PMC534705] [PubMed: 15537739]
- Sevelinges Y., Moriceau S., Holman P., Miner C., Muzny K., Gervais R., Mouly A.M., Sullivan R.M. Enduring effects of infant memories: Infant odor-shock conditioning attenuates amygdala activity and adult fear conditioning. Biological Psychiatry. 2007;62(10):1070–79. [PubMed: 17826749]
- Sevelinges Y., Sullivan R.M., Messaoudi B., Mouly A.M. Neonatal odor-shock conditioning alters the neural network involved in odor fear learning at adulthood. Learning & Memory. 2008;15(9):649–56. [PMC free article: PMC2632787] [PubMed: 18772252]
- Shair H.N., Masmela J.R., Brunelli S.A., Hofer M.A. Potentiation and inhibition of ultrasonic vocalization of rat pups: Regulation by social cues. Developmental Psychobiology. 1997;30(3):195–200. [PubMed: 9104550]
- Shepherd G.M. Perception without a thalamus how does olfaction do it? Neuron. 2005;46(2):166–68. [PubMed: 15848795]
- Shionoya K., Moriceau S., Lunday L., Miner C., Roth T.L., Sullivan R.M. Development switch in neural circuitry underlying odor-malaise learning. Learning & Memory. 2006;13(6):801–8. [PMC free article: PMC1783635] [PubMed: 17101877]
- Shipley M.T., Ennis M. Functional organization of olfactory system. Journal of Neurobiology. 1996;30(1):123–76. [PubMed: 8727988]
- Siegel S., Domjan M. Backward conditioning as an inhibitory procedure. Learning and Motivation. 1971;2:1–11.
- Sigurdsson T., Doyere V., Cain C.K., LeDoux J.E. Long-term potentiation in the amygdala: A cellular mechanism of fear learning and memory. Neuropharmacology. 2007;52(1):215–27. [PubMed: 16919687]
- Singh P.J., Tucker A.M., Hofer M.A. Effects of nasal ZnSO4 irrigation and olfactory bulbectomy on rat pups. Physiology & Behavior. 1976;17(3):373–82. [PubMed: 1013182]
- Smotherman W.P. Odor aversion learning by the rat fetus. Physiology & Behavior. 1982;29(5):769–71. [PubMed: 6296892]
- Spear N.E., Rudy J.W. Tests of the ontogeny of learning and memory: Issues, methods and results. In: Shair H.N., Barr G.A., editors. Developmental Psychobiology:New Methods and Changing Concepts. New York: Oxford University Press; 1991. pp. 84–113.
- Stanley W.E. Differential human handling as reinforcing events and as treatments influencing later social behavior in basenji puppies. Psychological Reports. 1962;10:775–88.
- Stanton M.E. Multiple memory systems, development and conditioning. Behavior Brain Research. 2000;110(1–2):25–37. [PubMed: 10802301]
- Stanton M.E., Fox G.D., Carter C.S. Ontogeny of the conditioned eyeblink response in rats: Acquisition or expression? Neuropharmacology. 1998;37(4–5):623–32. [PubMed: 9705002]
- Stanton M.E., Levine S. Inhibition of infant glucocorticoid stress response: Specific role of maternal cues. Developmental Psychobiology. 1990;23(5):411–26. [PubMed: 2253818]
- Stanton M.E., Wallstrom J., Levine S. Maternal contact inhibits pituitary-adrenal stress responses in preweanling rats. Developmental Psychobiology. 1987;20(2):131–45. [PubMed: 3582776]
- Stehouwer D.J., Campbell B.A. Habituation of the forelimb-withdrawal response in neonatal rats. Journal of Experimental Psychology Animal Behavior Process. 1978;4(2):104–19. [PubMed: 670888]
- Stern D.N., Fieve R.R., Neff N.H., Costa E. The effect of lithium chloride administration on brain and heart norepinephrine turnover rates. Psychopharmacologia. 1969;14(4):315–22. [PubMed: 5350631]
- Suchecki D., Rosenfeld P., Levine S. Maternal regulation of the hypothalamic-pituitary-adrenal axis in the infant rat: The roles of feeding and stroking. Brain Research Developmental Brain Research. 1993;75(2):185–92. [PubMed: 8261610]
- Sullivan R.M., Landers M., Yeaman B., Wilson D.A. Good memories of bad events in infancy. Nature. 2000;407(6800):38–39. [PMC free article: PMC9196994] [PubMed: 10993064]
- Sullivan R.M., Leon M. Early olfactory learning induces an enhanced olfactory bulb response in young rats. Brain Research. 1986;392(1–2):278–82. [PubMed: 3708381]
- Sullivan R.M., Stackenwalt G., Nasr F., Lemon C., Wilson D.A. Association of an odor with activation of olfactory bulb noradrenergic beta-receptors or locus coeruleus stimulation is sufficient to produce learned approach responses to that odor in neonatal rats. Behavioral Neuroscience. 2000;114(5):957–62. [PMC free article: PMC1885991] [PubMed: 11085610]
- Sullivan R.M., Wilson D.A. The locus coeruleus, norepinephrine, and memory in newborns. Brain Research Bulletin. 1994;35(5–6):467–72. [PubMed: 7859104]
- Sullivan R.M., Wilson D.A., Wong R., Correa A., Leon M. Modified behavioral and olfactory bulb responses to maternal odors in preweanling rats. Brain Research Developmental Brain Research. 1990;53(2):243–47. [PubMed: 2357798]
- Suomi S.J. Gene-environment interactions and the neurobiology of social conflict. Annals of the New York Academy of Sciences. 2003;1008:132–39. [PubMed: 14998879]
- Swanson L.W., Kohler C. Anatomical evidence for direct projections from the entorhinal area to the entire cortical mantle in the rat. Journal of Neuroscience. 1986;6(10):3010–23. [PMC free article: PMC6568776] [PubMed: 3020190]
- Swanson L.W., Petrovich G.D. What is the amygdala? Trends Neuroscience. 1998;21(8):323–31. [PubMed: 9720596]
- Takahashi L.K., Nakashima B.R., Hong H., Watanabe K. The smell of danger: A behavioral and neural analysis of predator odor-induced fear. Neuroscience Biobehavioral Review. 2005;29(8):1157–67. [PubMed: 16095694]
- Tanabe T., Yarita H., Iino M., Ooshima Y., Takagi S.F. An olfactory projection area in orbitofrontal cortex of the monkey. Journal of Neurophysiology. 1975;38(5):1269–83. [PubMed: 809549]
- Thompson B.L., Erickson K., Schulkin J., Rosen J.B. Corticosterone facilitates retention of contextually conditioned fear and increases CRH mRNA expression in the amygdala. Behavioral Brain Research. 2004;149(2):209–15. [PubMed: 15129783]
- Thompson J., Sullivan R.M., Wilson D.A. Developmental emergence of fear learning corresponds with changes in amygdala synaptic plasticity. Brain Research. 2008;1200:58–65. [PMC free article: PMC2291207] [PubMed: 18295751]
- Touzani K., Sclafani A. Critical role of amygdala in flavor but not taste preference learning in rats. European Journal of Neuroscience. 2005;22(7):1767–74. [PubMed: 16197517]
- Van Toller S., Kendal-Reed M. A possible protocognitive role for odor in human infant development. Brain Cognition. 1995;29(3):275–93. [PubMed: 8838386]
- Verwer R.W., Van Vulpen E.H., Van Uum J.F. Postnatal development of amygdaloid projections to the prefrontal cortex in the rat studied with retrograde and anterograde tracers. Journal of Comparative Neurology. 1996;376(1):75–96. [PubMed: 8946285]
- Walker C.D., Perrin M., Vale W., Rivier C. Ontogeny of the stress response in the rat: Role of the pituitary and the hypothalamus. Endocrinology. 1986;118(4):1445–51. [PubMed: 3004915]
- Walker D.L., Paschall G.Y., Davis M. Glutamate receptor antagonist infusions into the basolateral and medial amygdala reveal differential contributions to olfactory vs context fear conditioning and expression. Learning & Memory. 2005;12(2):120–29. [PMC free article: PMC1074329] [PubMed: 15774945]
- Walz A., Omura M., Mombaerts P. Development and topography of the lateral olfactory tract in the mouse: Imaging by genetically encoded and injected fluorescent markers. Journal of Neurobiology. 2006;66(8):835–46. [PubMed: 16673392]
- Wiedenmayer C.P., Barr G.A. Developmental changes in c-fos expression to an age-specific social stressor in infant rats. Behavior Brain Research. 2001;126(1–2):147–57. [PubMed: 11704260]
- Willander J., Larsson M. Smell your way back to childhood: Autobiographical odor memory. Psychonomic Bulletin & Reviews. 2006;13(2):240–44. [PubMed: 16892988]
- Willander J., Larsson M. Olfaction and emotion: The case of autobiographical memory. Memory Cognition. 2007;35(7):1659–63. [PubMed: 18062543]
- Wilson D.A. Habituation of odor responses in the rat anterior piriform cortex. Journal of Neurophysiology. 1998;79(3):1425–40. [PubMed: 9497422]
- Wilson D.A. Odor specificity of habituation in the rat anterior piriform cortex. Journal of Neurophysiology. 2000;83(1):139–45. [PubMed: 10634860]
- Wilson D.A., Stevenson R.J. Olfactory perceptual learning: The critical role of memory in odor discrimination. Neuroscience Biobehavioral Review. 2003;27(4):307–28. [PubMed: 12946684]
- Wilson D.A., Stevenson R.J. Learning to Smell:Olfactory Perception from Neurobiology to Behavior. Baltimore, MA: John Hopkins University; 2006.
- Wilson D.A., Sullivan R.M., Leon M. Single-unit analysis of postnatal olfactory learning: Modified olfactory bulb output response patterns to learned attractive odors. Journal of Neuroscience. 1987;7(10):3154–62. [PMC free article: PMC1892184] [PubMed: 3668621]
- Wilson R.C., Steward O. Polysynaptic activation of the dentate gyrus of the hippocampal formation: An olfactory input via the lateral entorhinal cortex. Experimental Brain Research. 1978;33(3–4):523–34. [PubMed: 215436]
- Witter M.P., Amaral D.G. Entorhinal cortex of the monkey: V. Projections to the dentate gyrus, hippocampus, and subicular complex. Journal of Comparative Neurology. 1991;307(3):437–59. [PubMed: 1713237]
- Woo C.C., Coopersmith R., Leon M. Localized changes in olfactory bulb morphology associated with early olfactory learning. Journal of Comparative Neurology. 1987;263(1):113–25. [PubMed: 3667967]
- Woo C.C., Oshita M.H., Leon M. A learned odor decreases the number of Fos-immunopositive granule cells in the olfactory bulb of young rats. Brain Research. 1996;716(1–2):149–56. [PubMed: 8738231]
- Yarita H., Iino M., Tanabe T., Kogure S., Takagi S.F. A transthalamic olfactory pathway to orbitofrontal cortex in the monkey. Journal of Neurophysiology. 1980;43(1):69–85. [PubMed: 6766180]
- Yuan Q., Harley C.W., Bruce J.C., Darby-King A., McLean J.H. Isoproterenol increases CREB phosphorylation and olfactory nerve-evoked potentials in normal and 5-HT-depleted olfactory bulbs in rat pups only at doses that produce odor preference learning. Learning & Memory. 2000;7(6):413–21. [PMC free article: PMC311343] [PubMed: 11112800]
- Memory and Plasticity in the Olfactory System: From Infancy to Adulthood - The N...Memory and Plasticity in the Olfactory System: From Infancy to Adulthood - The Neurobiology of Olfaction
- Antenatal and Postnatal Mental HealthAntenatal and Postnatal Mental Health
- Drug Class Review: Second-Generation AntidepressantsDrug Class Review: Second-Generation Antidepressants
- The Circulatory System and Oxygen Transport - Regulation of Tissue OxygenationThe Circulatory System and Oxygen Transport - Regulation of Tissue Oxygenation
- Learning from Pandemics Past - Ethical and Legal Considerations in Mitigating Pa...Learning from Pandemics Past - Ethical and Legal Considerations in Mitigating Pandemic Disease
Your browsing activity is empty.
Activity recording is turned off.
See more...
