NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-.

LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet].
Show detailsOVERVIEW
Introduction
Rufinamide is a unique anticonvulsant that is used in combination with other agents as therapy of severe forms of seizure disorders. Rufinamide therapy is associated with a low rate of transient serum enzyme elevations and with rare instances of clinically apparent liver injury.
Background
Rufinamide (roo fin' a mide) is a unique triazole derivative that appears to act by prolonging the inactivation of voltage gated sodium channels in the central nervous system, thus slowing the rate of neurotransmission and decreasing rapid, repetitive neuronal firing. Rufinamide was approved for use in the United States in 2008 as an anticonvulsant to be used in combination with other agents (adjunctive therapy) for Lennox-Gastaut syndrome in adults and children 1 year of age and older. Rufinamide is available in tablets of 200 and 400 mg and as an oral suspension of 40 mg/mL under the brand name Banzel. The typical dose in children is 10 mg/kg in two divided doses daily, which can be increased to a maximum of 45 mg/kg daily. In adults the dose is 400 to 800 mg daily in two divided doses, which can be increased to a maximum of 3200 mg daily. Side effects may include headache, dizziness, somnolence, ataxia, tremor, fatigue, nausea and rash. Rare, but potentially severe adverse events include depression, suicidal thoughts and behavior, mood changes and hypersensitivity reactions including Stevens Johnson syndrome.
Hepatotoxicity
In prelicensure clinical trials, addition of rufinamide to standard anticonvulsant therapy was reported to be associated with only rare elevations in ALT above 3 times the upper limit of normal (ULN). Rufinamide was not linked to instances of clinically apparent liver injury, but a pooled analysis of more than 200 children mentioned that two patients needed to discontinue therapy early because of liver related adverse events, one of which was described as “toxic hepatitis”. Since approval, there have been no reports of clinically apparent liver injury associated with rufinamide use, but it has had limited use in epilepsy. Rufinamide has been linked to instances of severe cutaneous reactions, including Stevens Johnson syndrome which often has some degree of associated liver injury. Thus, rufinamide may cause liver injury, but it is rare.
Likelihood score: E* (unproven but suspected cause of clinically apparent liver injury).
Mechanism of Injury
Rufinamide is metabolized by the liver, largely by CYP 2C19, but has not been reported to have significant drug interactions. The possible mechanism of hepatic injury from rufinamide is not known, but may relate to a toxic or immunogenic metabolite.
Outcome and Management
There is no information on the possible cross sensitivity to hepatotoxicity between rufinamide and other anticonvulsants, but its structure would not suggest that there would not be shared sensitivity with the more commonly used medications for seizures.
Drug Class: Anticonvulsants
PRODUCT INFORMATION
REPRESENTATIVE TRADE NAMES
Rufinamide – Banzel®
DRUG CLASS
Anticonvulsants
Product labeling at DailyMed, National Library of Medicine, NIH
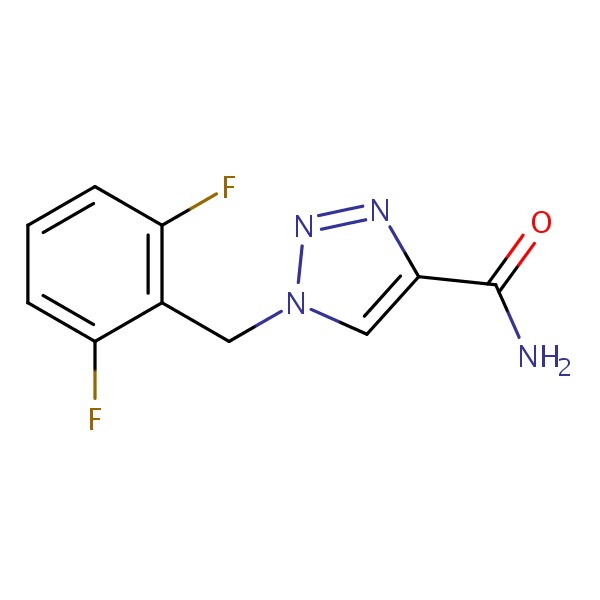
CHEMICAL FORMULA AND STRUCTURE
| DRUG | CAS REGISTRY NO. | MOLECULAR FORMULA | STRUCTURE |
|---|---|---|---|
| Rufinamide | 106308-44-5 | C10-H8-F2-N4-O |
 |
ANNOTATED BIBLIOGRAPHY
References updated: 06 June 2018
- Kaplowitz Zimmerman HJ. Anticonvulsants. In, Zimmerman, HJ. Hepatotoxicity: the adverse effects of drugs and other chemicals on the liver. 2nd ed. Philadelphia: Lippincott, 1999: pp. 498-516.(Expert review of anticonvulsants and liver injury published in 1999 before the availability of rufinamide).
- Pirmohamed M, Leeder SJ. Anticonvulsant agents. In, Kaplowitz N, DeLeve LD, eds. Drug-induced liver disease. 3rd ed. Amsterdam: Elsevier, 2013: pp 423-42.(Review of anticonvulsant induced liver injury; rufinamide is not discussed).
- McNamara JO. Rufinamide. Pharmacotherapy of the epilepsies. In, Brunton LL, Chabner BA, Knollman BC, eds. Goodman & Gilman’s the pharmacological basis of therapeutics. 12th ed. New York: McGraw-Hill, 2011, p. 602.(Textbook of pharmacology and therapeutics).
- Pålhagen S, Canger R, Henriksen O, van Parys JA, Rivière ME, Karolchyk MA. Rufinamide: a double-blind, placebo-controlled proof of principle trial in patients with epilepsy. Epilepsy Res. 2001; 43: 115-24. [PubMed: 11164700](Among 50 patients with seizures treated with rising doses of rufinamide or placebo for 28 days, side effects included fatigue, headache, tremor, ataxia and dizziness, but there were no “clinically relevant” changes in laboratory test results).
- Glauser T, Kluger G, Sachdeo R, Krauss G, Perdomo C, Arroyo S. Rufinamide for generalized seizures associated with Lennox-Gastaut syndrome. Neurology 2008; 70: 1950-8. [PubMed: 18401024](Among 138 patients with Lennox-Gastaut syndrome [ages 4 to 30 years] treated with rufinamide or placebo for 12 weeks, rufinamide was not associated with “clinically significant changes in … laboratory tests”).
- Wisniewski CS. Rufinamide: a new antiepileptic medication for the treatment of seizures associated with Lennox-Gastaut syndrome. Ann Pharmacother 2010; 44: 658-67. [PubMed: 20233912](Review of the efficacy and safety of rufinamide; no discussion of ALT elevations or hepatotoxicity).
- Rufinamide (Banzel) for epilepsy. Med Lett Drugs Ther 2009; 51 (1307): 18-20. [PubMed: 19265777](Concise review of the pharmacology, efficacy, adverse events, drug interactions and costs of rufinamide shortly after its approval in the US mentions common side effects, but not ALT elevations or hepatotoxicity).
- Brodie MJ, Rosenfeld WE, Vazquez B, Sachdeo R, Perdomo C, Mann A, Arroyo S. Rufinamide for the adjunctive treatment of partial seizures in adults and adolescents: a randomized placebo-controlled trial. Epilepsia 2009; 50: 1899-909. [PubMed: 19490053](Among 313 patients with partial onset seizures treated with rufinamide or placebo for 13 weeks, common side effects included dizziness, headache, nausea, somnolence and diplopia; no mention of ALT elevations or hepatotoxicity).
- Wheless JW, Conry J, Krauss G, Mann A, LoPresti A, Narurkar M. Safety and tolerability of rufinamide in children with epilepsy: a pooled analysis of 7 clinical studies. J Child Neurol 2009; 24: 1520-5. [PubMed: 19955344](In a pooled analysis of 7 clinical studies including 212 rufinamide and 197 placebo recipients, changes in laboratory values “were generally clinically insignificant”, however, there were two discontinuations for liver related adverse events, one for “toxic hepatitis” resolving within 17 days and one for serum enzyme elevations resolving within 4 days; 5 children developed a hypersensitivity reaction [fever and rash within the first 4 weeks of treatment], but hepatic involvement was not mentioned).
- Elger CE, Stefan H, Mann A, Narurkar M, Sun Y, Perdomo C. A 24-week multicenter, randomized, double-blind, parallel-group, dose-ranging study of rufinamide in adults and adolescents with inadequately controlled partial seizures. Epilepsy Res 2010; 88: 255-63. [PubMed: 20061123](Among 647 patients with poorly controlled partial onset seizures treated with adjunctive rufinamide [200, 400, 800 or 1600 mg daily] or placebo for 24 weeks, there were no liver related serious adverse events or early discontinuation; no mention of ALT elevations).
- Biton V, Krauss G, Vasquez-Santana B, Bibbiani F, Mann A, Perdomo C, Narurkar M. A randomized, double-blind, placebo-controlled, parallel-group study of rufinamide as adjunctive therapy for refractory partial-onset seizures. Epilepsia 2011; 52: 234-42. [PubMed: 20887365](Among 357 patients treated with adjunctive rufinamide or placebo for 3 months, there were no significant changes in laboratory values or discontinuations for liver related adverse events).
- Moavero R, Cusmai R, Specchio N, Fusco L, Capuano A, Curatolo P, Vigevano F. Rufinamide efficacy and safety as adjunctive treatment in children with focal drug resistant epilepsy: the first Italian prospective study. Epilepsy Res 2012; 102: 94-9. [PubMed: 22677424](Among 70 children with focal seizures treated with add on rufinamide for up to 12 months, the authors found no laboratory test abnormalities).
- Gaitatzis A, Sander JW. The long-term safety of antiepileptic drugs. CNS Drugs 2013; 27: 435-55. [PubMed: 23673774](Review of the long term safety and adverse event profile of anticonvulsants mentions that valproate and felbamate can cause liver failure, but does no mention hepatotoxicity of other antiepileptics).
- Drugs for epilepsy. Treat Guidel Med Lett 2013; 11: 9-18. [PubMed: 23348233](Concise review of indications and side effects of anticonvulsants; rufinamide is approved as add on therapy of Lennox-Gastaut syndrome in children and adults; discussion of adverse effects does not mention hepatotoxicity, but mentions that rufinamide is a mild inducer of CYP 3A4).
- Björnsson ES, Bergmann OM, Björnsson HK, Kvaran RB, Olafsson S. Incidence, presentation and outcomes in patients with drug-induced liver injury in the general population of Iceland. Gastroenterology 2013; 144: 1419-25. [PubMed: 23419359](In a population based study of drug induced liver injury from Iceland, 96 cases were identified over a 2 year period, but none were attributed to rufinamide).
- Grosso S, Coppola G, Dontin SD, Gobbi G, Pruna D, Accorsi P, Verrotti A, et al. Efficacy and safety of rufinamide in children under four years of age with drug-resistant epilepsies. Eur J Paediatr Neurol 2014; 18: 641-5. [PubMed: 24912730](Among 40 children with refractory epilepsy receiving add on rufinamide for an average of 12 months, “biochemical tests resulted normal in all patients”).
- Thome-Souza S, Kadish NE, Ramgopal S, Sánchez Fernández I, Bergin AM, Bolton J, Harini C, et al. Safety and retention rate of rufinamide in 300 patients: a single pediatric epilepsy center experience. Epilepsia 2014; 55: 1235-44. [PubMed: 25070475](Among 300 patients [ages 1 to 30 years] with refractory epilepsy receiving add on rufinamide for an average of 9 months, 110 [37%] discontinued therapy early, 47 [16%] for adverse events, but none for liver related side effects; no mention of ALT elevations or hepatotoxicity).
- Ohtsuka Y, Yoshinaga H, Shirasaka Y, Takayama R, Takano H, Iyoda K. Rufinamide as an adjunctive therapy for Lennox-Gastaut syndrome: a randomized double-blind placebo-controlled trial in Japan. Epilepsy Res 2014; 108: 1627-36. [PubMed: 25219353](Among 59 Japanese patients with Lennox-Gastaut syndrome receiving adjunctive rufinamide for partial onset seizures for 12 weeks, none had a serious adverse event or discontinuation for an adverse event; no mention of ALT elevations or hepatotoxicity).
- Chalasani N, Bonkovsky HL, Fontana R, Lee W, Stolz A, Talwalkar J, Reddy KR, et al.; United States Drug Induced Liver Injury Network. Features and outcomes of 899 patients with drug-induced liver injury: The DILIN Prospective Study. Gastroenterology 2015; 148: 1340-52. [PMC free article: PMC4446235] [PubMed: 25754159](Among 899 cases of drug induced liver injury enrolled in a US prospective study between 2004 and 2013, 40 [4.5%] were attributed to anticonvulsants, but none to rufinamide).
- McMurray R, Striano P. Treatment of Adults with Lennox-Gastaut Syndrome: Further analysis of efficacy and safety/tolerability of rufinamide. Neurol Ther 2016; 5: 35-43. [PMC free article: PMC4919131] [PubMed: 26861566](Among 31 adults with seizures treated with rufinamide or placebo for 84 days, adverse events were mild-to-moderate in severity and there were no "clinically significant changes" in laboratory tests).
- Ohtsuka Y, Yoshinaga H, Shirasaka Y, Takayama R, Takano H, Iyoda K. Long-term safety and seizure outcome in Japanese patients with Lennox-Gastaut syndrome receiving adjunctive rufinamide therapy: An open-label study following a randomized clinical trial. Epilepsy Res 2016; 121: 1-7. [PubMed: 26827266](Among 54 patients with Lennox-Gastaut syndrome treated for at least 52 weeks in a open label extension trial, seizure control was maintained and side effects included somnolence, anorexia and weight loss; no mention of ALT elevations or hepatotoxicity).
- Nikanorova M, Brandt C, Auvin S, McMurray R. Real-world data on rufinamide treatment in patients with Lennox-Gastaut syndrome: Results from a European noninterventional registry study. Epilepsy Behav 2017; 76: 63-70. [PubMed: 28927712](Among 64 patients followed in a prospective registry who started rufinamide treatment, most adverse events were mild-to-moderate in severity and included somnolence [8%], decreased appetite [6%] and fatigue [5%]; no mention of ALT elevations or hepatotoxicity).
- PMCPubMed Central citations
- PubChem SubstanceRelated PubChem Substances
- PubMedLinks to PubMed
- Rufinamide add-on therapy for drug-resistant epilepsy.[Cochrane Database Syst Rev. 2020]Rufinamide add-on therapy for drug-resistant epilepsy.Panebianco M, Prabhakar H, Marson AG. Cochrane Database Syst Rev. 2020 Nov 8; 11(11):CD011772. Epub 2020 Nov 8.
- Review Lacosamide.[LiverTox: Clinical and Researc...]Review Lacosamide.. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. 2012
- Review Rufinamide add-on therapy for refractory epilepsy.[Cochrane Database Syst Rev. 2018]Review Rufinamide add-on therapy for refractory epilepsy.Panebianco M, Prabhakar H, Marson AG. Cochrane Database Syst Rev. 2018 Apr 25; 4(4):CD011772. Epub 2018 Apr 25.
- The anticonvulsant profile of rufinamide (CGP 33101) in rodent seizure models.[Epilepsia. 2008]The anticonvulsant profile of rufinamide (CGP 33101) in rodent seizure models.White HS, Franklin MR, Kupferberg HJ, Schmutz M, Stables JP, Wolf HH. Epilepsia. 2008 Jul; 49(7):1213-20.
- Long-term safety and seizure outcome in Japanese patients with Lennox-Gastaut syndrome receiving adjunctive rufinamide therapy: An open-label study following a randomized clinical trial.[Epilepsy Res. 2016]Long-term safety and seizure outcome in Japanese patients with Lennox-Gastaut syndrome receiving adjunctive rufinamide therapy: An open-label study following a randomized clinical trial.Ohtsuka Y, Yoshinaga H, Shirasaka Y, Takayama R, Takano H, Iyoda K. Epilepsy Res. 2016 Mar; 121:1-7. Epub 2016 Jan 12.
- Rufinamide - LiverToxRufinamide - LiverTox
- Celecoxib - StatPearlsCelecoxib - StatPearls
- Hepatitis A - StatPearlsHepatitis A - StatPearls
- GDF10 growth differentiation factor 10 [Homo sapiens]GDF10 growth differentiation factor 10 [Homo sapiens]Gene ID:2662Gene
- 2662[uid] AND (alive[prop]) (1)Gene
Your browsing activity is empty.
Activity recording is turned off.
See more...