NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-.

LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet].
Show detailsOVERVIEW
Introduction
Palbociclib is a unique cyclin-dependent kinase inhibitor that is used in combination with aromatase inhibitors in the treatment of postmenopausal women with metastatic breast cancer. Palbociclib is associated with transient and usually mild elevations in serum aminotransferase during therapy and to an unusual form of liver injury called pseudocirrhosis caused by shrinkage of tumor metastases in the liver combined with desmoplastic changes and vascular damage, that can be severe, progressive and even fatal.
Background
Palbociclib (pal" boe sye' klib) is an orally available, specific inhibitor of cyclin-dependent kinases that is used in combination with aromatase inhibitors in the therapy of postmenopausal women with metastatic breast cancer that is positive for the estrogen receptor (ER+), but negative for human epidermal growth factor receptor 2 (HER2-). The cyclin kinases 4 and 6 regulate the cellular transition from the G1 to the S phase of the cell cycle. Inhibition of this transition blocks the progression of the cell cycle and results in growth arrest in rapidly dividing cells. The addition of palbociclib to letrozole or fulvestrant (aromatase inhibitors) therapy of metastatic breast cancer (ER+, HER2-) in postmenopausal women was associated with a prolongation of disease free survival. Palbociclib received accelerated approval for use in the United States in 2015, and it is still under close evaluation for its long term safety and efficacy. Palbociclib is available in capsules of 75, 100 and 125 mg and the typical maintenance dose is 125 mg once daily in 21 day cycles every 28 days indefinitely or until there is disease progression. Common side effects include fatigue, nausea, diarrhea, anorexia, neutropenia, fever, anemia, thrombocytopenia, epistasis, peripheral neuropathy and pulmonary embolism. Severe adverse events include neutropenic fever and sepsis.
Hepatotoxicity
In the large clinical trials, adverse events were common and led to dose reductions in one-third of patients and discontinuation in 8%. Publications on the efficacy and safety of palbociclib rarely mentioned serum ALT elevations or hepatotoxicity. In a study of women with refractory, metastatic breast cancer, serum ALT elevations occurred in 6% [2% over 5 times ULN] receiving palbociclib and fulvestrant compared to 3% [none over 5 times ULN] on fulvestrant alone. Since its approval and more widescale use, there have been several reports of prominent ALT elevations arising after 2 or 3 cycles of palbociclib, that improved on discontinuation and recurred rapidly when restarted. Serum bilirubin and alkaline phosphatase levels were normal and symptoms were not mentioned. In addition, there have been rare reports of patients with refractory metastatic breast cancer who developed pseudocirrhosis within 2 to 3 months of starting palbociclib presenting with fatigue, jaundice and ascites with only modest elevations in serum aminotransferase and alkaline phosphatase levels. Imaging revealed a severely nodular liver, but liver histology showed desmoplastic changes in areas of necrotic metastatic tumor without cirrhosis. The liver also had vascular changes suggestive of sinusoidal obstruction syndrome, changes possibly caused by the dramatic involution of the metastatic tumor tissue combined with vascular damage. Pseudocirrhosis has been reported with other highly successful antineoplastic therapies of cancer metastatic to the liver, but the frequency is rare.
Likelihood score: C (probable rare cause of clinically apparent liver injury that may represent pseudocirrhosis from nodular transformation of the liver in response to necrosis of hepatic metastases).
Mechanism of Injury
The possible causes of serum enzyme elevations or liver injury from palbociclib therapy are not known. Palbociclib is metabolized in the liver largely through the CYP 3A4 pathway and liver injury might be caused by production of a toxic or immunogenic intermediate. Because it is a substrate for CYP 3A4, palbociclib is susceptible to drug-drug interactions with agents that inhibit or induce this specific hepatic microsomal activity. Pseudocirrhosis due to palbociclib is thought to be due to the necrosis and involution of hepatic metastases that cause an altered hepatic architecture and vasculature resulting in liver dysfunction.
Outcome and Management
Serum aminotransferase elevations above 5 times the upper limit of normal (if confirmed) or any elevations accompanied by jaundice or symptoms should lead to dose reduction or temporary cessation. There is no evidence to suggest a cross reactivity in risk for adverse events, hypersensitivity or hepatic injury between palbociclib and other cyclin dependent kinase inhibitors.
Drug Class: Antineoplastic Agents, Protein Kinase Inhibitors
Other Cyclin-Dependent Kinase Inhibitor Drugs: Ribociclib
PRODUCT INFORMATION
REPRESENTATIVE TRADE NAMES
Palbociclib – Ibrance®
DRUG CLASS
Antineoplastic Agents
Product labeling at DailyMed, National Library of Medicine, NIH
CHEMICAL FORMULA AND STRUCTURE
| DRUG | CAS REGISTRY NO. | MOLECULAR FORMULA | STRUCTURE |
|---|---|---|---|
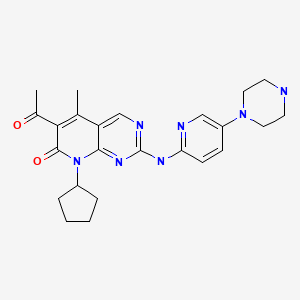
| Palbociclib | 571190-30-2 | C24-H29-N7-O2 |
|
ANNOTATED BIBLIOGRAPHY
References updated: 10 April 2020
Abbreviations: ER, estrogen receptor; HER2, human epidermal growth factor receptor 2.
- Zimmerman HJ. Zimmerman HJ. Hepatotoxicity: the adverse effects of drugs and other chemicals on the liver. 2nd ed. Philadelphia: Lippincott, 1999.(Review of hepatotoxicity published in 1999 before the availability of tyrosine kinase inhibitors such as palbociclib).
- DeLeve LD. Erlotinib. Cancer chemotherapy. In, Kaplowitz N, DeLeve LD, eds. Drug-induced liver disease. 3rd ed. Amsterdam: Elsevier, 2013, pp. 556.(Review of hepatotoxicity of cancer chemotherapeutic agents discusses several tyrosine kinase inhibitors including imatinib, gefitinib, erlotinib and crizotinib, but not palbociclib).
- Wellstein A, Giaccone G, Atkins MB, Sausville EA. Pathway-targeted therapies: monoclonal antibodies, protein kinase inhibitors, and various small molecules. In, Brunton LL, Hilal-Dandan R, Knollman BC, eds. Goodman & Gilman's the pharmacological basis of therapeutics. 13th ed. New York: McGraw-Hill, 2018, pp. 1203-36.(Textbook of pharmacology and therapeutics).
- Spraggs CF, Xu CF, Hunt CM. Genetic characterization to improve interpretation and clinical management of hepatotoxicity caused by tyrosine kinase inhibitors. Pharmacogenomics. 2013;14:541–54. [PubMed: 23556451](Review of genetic associations of serum ALT and bilirubin elevations during therapy with tyrosine kinase inhibitors focusing on lapatinib and pazopanib; palbociclib is not discussed).
- Shah RR, Morganroth J, Shah DR. Hepatotoxicity of tyrosine kinase inhibitors: clinical and regulatory perspectives. Drug Saf. 2013;36:491–503. [PubMed: 23620168](Review of the hepatotoxicity of 18 tyrosine kinase inhibitors approved for use in cancer in the US as of 2013, before the availability of palbociclib which is not discussed).
- Dickson MA, Tap WD, Keohan ML, D'Angelo SP, Gounder MM, Antonescu CR, Landa J, et al. Phase II trial of the CDK4 inhibitor PD0332991 in patients with advanced CDK4-amplified well-differentiated or dedifferentiated liposarcoma. J Clin Oncol. 2013;31:2024–8. [PMC free article: PMC3661937] [PubMed: 23569312](Among 30 patients with liposarcoma treated with palbociclib adverse events included neutropenia [50%], thrombocytopenia [30%] and anemia [17%], with no mention of ALT elevations or hepatotoxicity).
- Vaughn DJ, Hwang WT, Lal P, Rosen MA, Gallagher M, O'Dwyer PJ. Phase 2 trial of the cyclin-dependent kinase 4/6 inhibitor palbociclib in patients with retinoblastoma protein-expressing germ cell tumors. Cancer. 2015;121:1463–8. [PubMed: 25522918](Among 30 patients with germ cell tumors treated with palbociclib, response rates were poor and toxicities were largely hematologic; no mention of ALT elevations or hepatotoxicity).
- Beaver JA, Amiri-Kordestani L, Charlab R, Chen W, Palmby T, Tilley A, Zirkelbach JF, et al. FDA Approval: Palbociclib for the treatment of postmenopausal patients with estrogen receptor-positive, HER2-negative metastatic breast cancer. Clin Cancer Res. 2015;21:4760–6. [PubMed: 26324739](Review of the studies of clinical efficacy and safety that led to the FDA approval of palbociclib; mentions that adverse events were common and led to dose reduction in 36% and discontinuation in 8% of patients; no mention of ALT elevations or hepatotoxicity).
- Mangini NS, Wesolowski R, Ramaswamy B, Lustberg MB, Berger MJ. Palbociclib: a novel cyclin-dependent kinase inhibitor for hormone receptor-positive advanced breast cancer. Ann Pharmacother. 2015;49:1252–60. [PMC free article: PMC7331461] [PubMed: 26324355](Review of the structure, mechanism of action, pharmacology, clinical efficacy and safety of palbociclib as therapy of metastatic breast cancer, discusses neutropenia and infections but does not mention ALT elevations or hepatotoxicity).
- Finn RS, Crown JP, Lang I, Boer K, Bondarenko IM, Kulyk SO, Ettl J, et al. The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study. Lancet Oncol. 2015;16:25–35. [PubMed: 25524798](Among 165 women with metastatic breast cancer [ER+, HER2-] treated with letrozole with or without palbociclib for an average of 28 months, progression-free survival was longed with palbociclib, but side effects including neutropenia were more frequent and severe; no mention of ALT elevations or hepatotoxicity).
- Turner NC, Ro J, André F, Loi S, Verma S, Iwata H, Harbeck N, et al. PALOMA3 Study Group. Palbociclib in Hormone-Receptor-Positive Advanced Breast Cancer. N Engl J Med. 2015;373:209–19. [PubMed: 26030518](Among 521 women with advanced breast cancer [ER+, HER2-] treated with letrozole and either palbociclib or placebo for up to 1 year, progression-free survival was prolonged by adding palbociclib [9.2 vs 3.8 months] and side effects were more common including neutropenia [62% vs 1%] and fatigue [38% vs 26%]; no mention of ALT elevations or hepatotoxicity).
- DeMichele A, Clark AS, Tan KS, Heitjan DF, Gramlich K, Gallagher M, Lal P, et al. CDK 4/6 inhibitor palbociclib (PD0332991) in Rb+ advanced breast cancer: phase II activity, safety, and predictive biomarker assessment. Clin Cancer Res. 2015;21:995–1001. [PubMed: 25501126](Among 37 women with metastatic breast cancer [ER+, HER2-] treated with palbociclib, adverse events [largely hematologic] were common, but rarely led to dose interruption; no mention of ALT elevations or hepatotoxicity).
- Palbociclib (Ibrance) for metastatic breast cancer. Med Lett Drugs Ther. 2015;57(1475):115–6. [PubMed: 26262882](Concise review of efficacy and safety of palbociclib as therapy for metastatic breast cancer, mentions frequency of severe neutropenia and risk of septicemia; no mention of hepatotoxicity or ALT elevations).
- Cristofanilli M, Turner NC, Bondarenko I, Ro J, Im SA, Masuda N, Colleoni M, et al. Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2-negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): final analysis of the multicentre, double-blind, phase 3 randomised controlled trial. Lancet Oncol. 2016;17:425–39. [PubMed: 26947331](Among 521 women with refractory, metastatic ER+, HER2- breast cancer treated with fulvestrant with or without palbociclib, progression-free survival was improved with palbociclib therapy [9.5 vs 4.6 months] but adverse events were more frequent including neutropenia [65% vs 1%], while ALT elevations arose in 6% vs 3% and were above 5 times ULN in 2% vs 0%]).
- Finn RS, Martin M, Rugo HS, Jones S, Im SA, Gelmon K, Harbeck N, et al. Palbociclib and letrozole in advanced breast cancer. N Engl J Med. 2016;375(20):1925–36. [PubMed: 27959613](Among 666 women with previously untreated, ER+, HER2- metastatic breast cancer treated with letrozole with or without palbociclib, the median progression-free survival was greater with palbociclib [24.8 vs 14.5 months], but neutropenia was common [66% vs 1%], no mention of ALT elevations or hepatotoxicity).
- Vuppalanchi R, Saxena R, Storniolo AMV, Chalasani N. Pseudocirrhosis and liver failure in patients with metastatic breast cancer after treatment with palbociclib. Hepatology. 2017;65:1762–4. [PubMed: 27397671](Two women, ages 47 and 61 years with refractory metastatic breast cancer developed fatigue, jaundice and ascites after 3 and 2 cycles of palbociclib [bilirubin 11.7 and 2.6 mg/dL rising to 34 and 10 mg/dL, ALT 59 and 53 U/L, Alk P 128 and 243 U/L], imaging studies showing a nodular liver and ascites but liver histology showing pseudocirrhosis and possibly sinusoidal obstruction syndrome, both patients dying of hepatic failure).
- Roberts BA, Ibrahim M, Stone E. Elevated liver function tests in a patient on palbociclib and fulvestrant. J Community Support Oncol. 2018;16:e277–e279.(57 year old woman with metastatic breast cancer developed abnormal liver tests after 3 monthly cycles of palbociclib and fulvestrant [ALT 446 U/L, bilirubin and Alk P normal], which resolved within 4-6 weeks despite restarting fulvestrant but then recurred within 10 days of restarting palbociclib [ALT 285 U/L]).
- Ettl J. Management of adverse events due to cyclin-dependent kinase 4/6 inhibitors. Breast Care (Basel). 2019;14(2):86–92. [PMC free article: PMC6886109] [PubMed: 31798379](Overview recommendations on management of adverse events associated with cyclin-dependent kinase 4/5 inhibitors focusing upon neutropenia, mentions that monitoring of routine liver tests is recommended on days 1 and 14 of the first 2 cycles of therapy and on day 1 of subsequent courses, but that ALT elevations are more frequent with ribociclib and abemaciclib than with palbociclib, and there does not appear to be cross sensitivity to the hepatic adverse effects).
- Farhat F, Tarabaih M, Kanj A, Aoun M, Kattan J, Assi T, Awada A. Palbociclib safety and efficacy beyond ribociclib-induced liver toxicity in metastatic hormone-receptors positive breast cancer patient. Anticancer Drugs. 2020;31:85–9. [PubMed: 31609762](55 year old woman with metastatic breast cancer had a clinical response to ribociclib therapy but after 3 months developed ALT elevations [424 U/L], which improved on stopping but rose again upon restarting (ALT 613 U/L), yet later tolerated palbociclib therapy and maintained normal ALT levels).
- Atallah R, Parker NA, Hamouche K, Truong QV, Dingwall M. Palbociclib-induced liver failure. Kans J Med. 2020;13:81–2. [PMC free article: PMC7182058] [PubMed: 32337006](79 year old woman with metastatic breast cancer was found to have abnormal liver tests after a second course of palbociclib and fulvestrant [bilirubin 4.6 rising to 9.8 mg/dL, ALT 50 U/L, Alk P 150 U/L, INR 3.8], with E. coli septicemia and progressive multiorgan failure and death).
- PMCPubMed Central citations
- PubChem SubstanceRelated PubChem Substances
- PubMedLinks to PubMed
- Review Differences of cyclin-dependent kinase 4/6 inhibitor, palbociclib and abemaciclib, in breast cancer.[Jpn J Clin Oncol. 2019]Review Differences of cyclin-dependent kinase 4/6 inhibitor, palbociclib and abemaciclib, in breast cancer.Tamura K. Jpn J Clin Oncol. 2019 Dec 18; 49(11):993-998.
- The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study.[Lancet Oncol. 2015]The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study.Finn RS, Crown JP, Lang I, Boer K, Bondarenko IM, Kulyk SO, Ettl J, Patel R, Pinter T, Schmidt M, et al. Lancet Oncol. 2015 Jan; 16(1):25-35. Epub 2014 Dec 16.
- Real-World Tumor Response of Palbociclib in Combination With an Aromatase Inhibitor as First-Line Therapy in Pre/Perimenopausal Women With Metastatic Breast Cancer.[Target Oncol. 2023]Real-World Tumor Response of Palbociclib in Combination With an Aromatase Inhibitor as First-Line Therapy in Pre/Perimenopausal Women With Metastatic Breast Cancer.DeMichele A, Robert N, Chen C, Kim S, Zhang Z, Lu DR, Aguilar KM, Wang Y, Li B, Schneeweiss S, et al. Target Oncol. 2023 Jul; 18(4):543-558. Epub 2023 Jul 10.
- Review Role of cyclin-dependent kinase 4/6 inhibitors in the current and future eras of cancer treatment.[J Oncol Pharm Pract. 2019]Review Role of cyclin-dependent kinase 4/6 inhibitors in the current and future eras of cancer treatment.Parylo S, Vennepureddy A, Dhar V, Patibandla P, Sokoloff A. J Oncol Pharm Pract. 2019 Jan; 25(1):110-129. Epub 2018 May 4.
- Treatment patterns and clinical outcomes among patients receiving palbociclib in combination with an aromatase inhibitor or fulvestrant for HR+/HER2-negative advanced/metastatic breast cancer in real-world settings in the US: Results from the IRIS study.[Breast. 2019]Treatment patterns and clinical outcomes among patients receiving palbociclib in combination with an aromatase inhibitor or fulvestrant for HR+/HER2-negative advanced/metastatic breast cancer in real-world settings in the US: Results from the IRIS study.Taylor-Stokes G, Mitra D, Waller J, Gibson K, Milligan G, Iyer S. Breast. 2019 Feb; 43:22-27. Epub 2018 Oct 20.
- Palbociclib - LiverToxPalbociclib - LiverTox
- Eczema: Learn More – Light therapy, tablets and injections - InformedHealth.orgEczema: Learn More – Light therapy, tablets and injections - InformedHealth.org
- Respiratory Diseases - The Health Consequences of Smoking—50 Years of ProgressRespiratory Diseases - The Health Consequences of Smoking—50 Years of Progress
- TPT1 tumor protein, translationally-controlled 1 [Homo sapiens]TPT1 tumor protein, translationally-controlled 1 [Homo sapiens]Gene ID:7178Gene
- 7178[uid] AND (alive[prop]) (1)Gene
Your browsing activity is empty.
Activity recording is turned off.
See more...