This book is distributed under the terms of the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0) ( http://creativecommons.org/licenses/by-nc-nd/4.0/ ), which permits others to distribute the work, provided that the article is not altered or used commercially. You are not required to obtain permission to distribute this article, provided that you credit the author and journal.
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-.

StatPearls [Internet].
Show detailsContinuing Education Activity
Striae, or stretch marks, are indented streaks that often affect the abdomen, buttocks, thighs, back, breasts, axillae and groin. They are divided into striae atrophicans (thinned skin), striae gravidarum (following pregnancy), striae distensae (stretched skin), striae rubrae (red), striae albae (white), striae nigra (black), and striae caerulea (dark blue). These common complaints are difficult to treat. This activity outlines the role of the interprofessional team in the evaluation and management of stretch marks.
Objectives:
- Review the causes of stretch marks.
- Describe the pathophysiology of stretch marks.
- Summarize the treatment options for stretech marks
- Explain the value of improving care coordination among the members of the interprofessional team to educate patients with stretch marks.
Introduction
Striae, or stretch marks, are a common complaint and can be distressing. They may affect the abdomen, buttocks, thighs, breasts, back, axillae and groin. They are classified according to appearance or epidemiology, as:
- striae atrophicans (thinned skin)
- striae gravidarum (following pregnancy)
- striae distensae (stretched skin)
- striae rubrae (red), striae albae (white)
- striae nigra (black)
- striae caerulea (dark blue).
Etiology
Striae are a form of dermal scarring associated with stretching of the dermis. They often result from a rapid change in weight (gain and loss) or are associated with endogenous or exogenous corticosteroids. Proposed mechanisms relate to hormones, physical stretch, and structural alterations of dermal collagen and elastic tissue. Adrenocorticotropic hormones promote fibroblast activity and increase protein catabolism. Pregnancy-related hormones may also contribute. Serum relaxin has been described to be lower in women with striae distensae.[3] Deficiency of fibrillin has also been proposed. [4] Genetic factors are unexplored, except that decreased expression of collagen and fibronectin genes is reported to be associated with striae.
Epidemiology
Striae distensae occur in pregnancy (43% to 88%), puberty (6% to 86%) and obesity (43%). Striae atrophicans follow medical conditions, particularly Cushing syndrome/disease, and treatments, usually exogenous topical or systemic corticosteroids [5], or surgery. Other associated diseases are Marfan syndrome[6], anorexia nervosa[7], various febrile illnesses, and chronic liver disease. Medications associated with striae also include chemotherapy, prolonged antibiotic therapy, contraceptives[8], and neuroleptics.
Striae are more common in females than in males and may be more common in certain races. They can appear more prominent in dark-skinned individuals. A positive family history is a risk factor for striae. During pregnancy, striae are more common in younger women than in older women. Several studies have noted greater prevalence with large abdominal circumference and large weight gain (due to fetal size or polyhydramnios). [9] One study reported that striae were more prevalent in smokers than non-smokers.
Pathophysiology
Pathophysiology is thought to involve elastases released from mast cells and macrophage activity. [10] Elastolysis of the mid-dermis is followed by a reorganization of collagen and fibrillin.
Histopathology of striae rubrae reveals excessive fine elastic fibers in the papillary dermis with thicker tortuous fibers in the periphery, with perivascular lymphocytes, dilated dermal vessels and edema. There are reduction and reorganization of elastin and fibrillin fibers, and structural changes in collagen fibers, which are thicker and densely packed in parallel rows. Histopathology of striae albae shows epidermal atrophy, loss of rete ridges, less vascularity, and densely packed, thin and scar-like horizontal collagen bundles.[4] They appear similar to mature atrophic scars.
Electron microscopy studies have also reported mast cell degranulation, macrophage activation, and elastolysis of mid dermis.[11]
History and Physical
History may explain the appearance of striae by association with stretched skin, for example, in pregnancy, pubertal growth spurt, muscular exercise, or weight gain. Inquire about topical application of potent corticosteroid or prolonged systemic steroids during the recent or remote past. If striae are widespread and none of these explanations are relevant, take a full medical history and conduct a thorough clinical examination.[12]
The initial striae rubrae are slightly raised pink or violaceous linear marks (striae rubrae), which fade over months to years to hypopigmented, atrophic, wrinkled scars (striae albae). The marks are perpendicular to the direction of skin tension. They fade with time. In pregnancy, they occur on the abdomen, breasts, and thighs. In adolescents, they are common on thighs, buttocks, breasts (females) and back (males).
Striae rubrae are sometimes pruritic. Otherwise, striae are asymptomatic. Treatment is sought because of their unsightly appearance.
Evaluation
Authors have used various methods of assessing the type and severity of striae when discussing the efficacy of treatments. These are not standardized or validated. Dermoscopy shows increased melanization in striae rubrae and reduced melanization in striae albae. [13] A biopsy is not necessary or useful.
Treatment / Management
The aim of treatment is to reduce redness, swelling and irritation in striae rubrae, and to increase collagen and elastic fiber production, improve hydration, and reduce inflammation in striae albae.[14]
Topical management is commonly recommended to prevent and treat striae, with very little if any evidence of efficacy, according to several published comprehensive reviews. Clinical trials have been of low quality involving small numbers of subjects.
- Many emollients and over-the-counter cosmeceuticals are marketed and used by pregnant women with the intention of preventing striae distensae or reducing their severity, resulting in considerable effort and expense, despite the considerable uncertainty whether any have benefit. [15]
- Silicone gels are recommended for atrophic scars and may be used in striae distensae. Published results are difficult to interpret.[16]
- Chemical peels using various acids have been used to treat striae. Efficacy is unknown.
Physical treatments are also advocated but also have little evidence to support their use.
- Massage is a component of topical therapy, and it is also used in scar management. [19]
- Exposure to broadband ultraviolet radiation can cause repigmentation in striae alba but is reported to have temporary effect with loss of pigmentation within a few months.
- Light and laser therapies have shown improvements in the appearance of striae, although it is uncertain which is best, and at what stage (rubrae, albae) they should be used. [19] Several laser types (e.g., pulse dye laser) target vascular chromophores in striae rubra and are reported to reduce redness and swelling.[20] Fractional lasers (e.g., Erbium-YAG) stimulate fibroblasts to produce collagen and elastin and can cause repigmentation in striae alba.[21]
- Light and lasers cause short-term erythema and oedema. They should be avoided in darker skin types due to greater risk of adverse effects, particularly transient postinflammatory hyperpigmentation and persistent hypopigmentation. Non-ablative devices are safer than ablative lasers.
- Radiofrequency energy devices emit high-frequency alternating electric current producing dermal heat. They are used to tighten the dermis, reduce wrinkles, and improve cellulite. Treatment promotes neocollagenesis, neoelastogenesis, and increased ground substances (proteoglycans) and should theoretically improve the appearance of striae. At least one published study has reported treatment was well tolerated, and the patients were satisfied with the results. A more recent advance has been the delivery of radiofrequency energy to a depth of 3.5 mm using a multiple needle delivery mode. Anecdotal reports are encouraging but proper studies are awaited. [22]
The effect of treatments is difficult to assess. For example, laser protocols use differing devices, fluence, pulse duration, spot size and treatment frequency and number. The utility of combination strategies is unknown.[14]
Platelet-rich plasma injections are also under investigation.[27]
When using any of these modalities, it is important to have standardized pre-treatment and post-treatment photographs. Furthermore, it is important to take these photographs six and twelve months out after the course of treatments. Many published reports do not have standardized photographs, nor do they have long-term follow up.
Differential Diagnosis
- Anetoderma
- Cutis Laxa
- Linear Focal Elastosis
- Mid-Dermal Elastosis
- Pseudoxanthoma Elasticum
- Striae from Topical Steroidal Abuse
Enhancing Healthcare Team Outcomes
Over time, many striae improve. The best time to perform a treatment (with proper photographs as discussed) is once the striae have stabilized. Many creams for striae are promoted on the internet with little evidence of the efficacy. The public should be made aware that the efficacy is very limited Newer lasers and radiofrequency treatments hold promise as they do try to modify the vascularity and the collagen and perhaps the elastin. Again, there is no clear leader among the many lasers that are promoted. The needle radiofrequency seems to show promise as, in theory, the deeper deliver of the energy may allow "shrinkage" of the striae in all directions, thereby improving the overall appearance. (Level V).
Review Questions
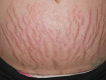
Figure
Striae Gravidarum Rubrae DermNet New Zealand
References
- 1.
- Farahnik B, Park K, Kroumpouzos G, Murase J. Striae gravidarum: Risk factors, prevention, and management. Int J Womens Dermatol. 2017 Jun;3(2):77-85. [PMC free article: PMC5440454] [PubMed: 28560300]
- 2.
- Al-Himdani S, Ud-Din S, Gilmore S, Bayat A. Striae distensae: a comprehensive review and evidence-based evaluation of prophylaxis and treatment. Br J Dermatol. 2014 Mar;170(3):527-47. [PubMed: 24125059]
- 3.
- Lurie S, Matas Z, Fux A, Golan A, Sadan O. Association of serum relaxin with striae gravidarum in pregnant women. Arch Gynecol Obstet. 2011 Feb;283(2):219-22. [PubMed: 20047054]
- 4.
- Wang F, Calderone K, Smith NR, Do TT, Helfrich YR, Johnson TR, Kang S, Voorhees JJ, Fisher GJ. Marked disruption and aberrant regulation of elastic fibres in early striae gravidarum. Br J Dermatol. 2015 Dec;173(6):1420-30. [PubMed: 26179468]
- 5.
- Neve S, Kirtschig G. Elastotic striae associated with striae distensae after application of very potent topical corticosteroids. Clin Exp Dermatol. 2006 May;31(3):461-2. [PubMed: 16681607]
- 6.
- Ledoux M, Beauchet A, Fermanian C, Boileau C, Jondeau G, Saiag P. A case-control study of cutaneous signs in adult patients with Marfan disease: diagnostic value of striae. J Am Acad Dermatol. 2011 Feb;64(2):290-5. [PubMed: 21112669]
- 7.
- Strumia R. Skin signs in anorexia nervosa. Dermatoendocrinol. 2009 Sep;1(5):268-70. [PMC free article: PMC2836432] [PubMed: 20808514]
- 8.
- Gupta M. Medroxyprogesterone acetate [Depo Provera] injections. Development of striae. Br J Fam Plann. 2000 Apr;26(2):104-5. [PubMed: 10773604]
- 9.
- Picard D, Sellier S, Houivet E, Marpeau L, Fournet P, Thobois B, Bénichou J, Joly P. Incidence and risk factors for striae gravidarum. J Am Acad Dermatol. 2015 Oct;73(4):699-700. [PubMed: 26369842]
- 10.
- Sheu HM, Yu HS, Chang CH. Mast cell degranulation and elastolysis in the early stage of striae distensae. J Cutan Pathol. 1991 Dec;18(6):410-6. [PubMed: 1774350]
- 11.
- Zheng P, Lavker RM, Kligman AM. Anatomy of striae. Br J Dermatol. 1985 Feb;112(2):185-93. [PubMed: 3970840]
- 12.
- Kasielska-Trojan A, Sobczak M, Antoszewski B. Risk factors of striae gravidarum. Int J Cosmet Sci. 2015 Apr;37(2):236-40. [PubMed: 25440082]
- 13.
- Hermanns JF, Piérard GE. High-resolution epiluminescence colorimetry of striae distensae. J Eur Acad Dermatol Venereol. 2006 Mar;20(3):282-7. [PubMed: 16503888]
- 14.
- Forbat E, Al-Niaimi F. Treatment of striae distensae: An evidence-based approach. J Cosmet Laser Ther. 2019;21(1):49-57. [PubMed: 29451986]
- 15.
- Rawlings AV, Bielfeldt S, Lombard KJ. A review of the effects of moisturizers on the appearance of scars and striae. Int J Cosmet Sci. 2012 Dec;34(6):519-24. [PubMed: 22994859]
- 16.
- Ud-Din S, McAnelly SL, Bowring A, Whiteside S, Morris J, Chaudhry I, Bayat A. A double-blind controlled clinical trial assessing the effect of topical gels on striae distensae (stretch marks): a non-invasive imaging, morphological and immunohistochemical study. Arch Dermatol Res. 2013 Sep;305(7):603-17. [PubMed: 23579949]
- 17.
- Ud-Din S, McGeorge D, Bayat A. Topical management of striae distensae (stretch marks): prevention and therapy of striae rubrae and albae. J Eur Acad Dermatol Venereol. 2016 Feb;30(2):211-22. [PMC free article: PMC5057295] [PubMed: 26486318]
- 18.
- Korgavkar K, Wang F. Stretch marks during pregnancy: a review of topical prevention. Br J Dermatol. 2015 Mar;172(3):606-15. [PubMed: 25255817]
- 19.
- Timur Taşhan S, Kafkasli A. The effect of bitter almond oil and massaging on striae gravidarum in primiparaous women. J Clin Nurs. 2012 Jun;21(11-12):1570-6. [PubMed: 22594386]
- 20.
- Elsaie ML, Hussein MS, Tawfik AA, Emam HM, Badawi MA, Fawzy MM, Shokeir HA. Comparison of the effectiveness of two fluences using long-pulsed Nd:YAG laser in the treatment of striae distensae. Histological and morphometric evaluation. Lasers Med Sci. 2016 Dec;31(9):1845-1853. [PubMed: 27595152]
- 21.
- Zaleski-Larsen LA, Jones IT, Guiha I, Wu DC, Goldman MP. A Comparison Study of the Nonablative Fractional 1565-nm Er: glass and the Picosecond Fractional 1064/532-nm Nd: YAG Lasers in the Treatment of Striae Alba: A Split Body Double-Blinded Trial. Dermatol Surg. 2018 Oct;44(10):1311-1316. [PubMed: 29746426]
- 22.
- Dover JS, Rothaus K, Gold MH. Evaluation of safety and patient subjective efficacy of using radiofrequency and pulsed magnetic fields for the treatment of striae (stretch marks). J Clin Aesthet Dermatol. 2014 Sep;7(9):30-3. [PMC free article: PMC4174917] [PubMed: 25276274]
- 23.
- Hersant B, Niddam J, Meningaud JP. Comparison between the efficacy and safety of platelet-rich plasma vs microdermabrasion in the treatment of striae distensae: clinical and histopathological study. J Cosmet Dermatol. 2016 Dec;15(4):565. [PubMed: 27320781]
- 24.
- Bitencourt S, Lunardelli A, Amaral RH, Dias HB, Boschi ES, de Oliveira JR. Safety and patient subjective efficacy of using galvanopuncture for the treatment of striae distensae. J Cosmet Dermatol. 2016 Dec;15(4):393-398. [PubMed: 27090205]
- 25.
- Aust M, Walezko N. [Acne scars and striae distensae: Effective treatment with medical skin needling]. Hautarzt. 2015 Oct;66(10):748-52. [PubMed: 26251169]
- 26.
- Kravvas G, Veitch D, Al-Niaimi F. The use of energy devices in the treatment of striae: a systematic literature review. J Dermatolog Treat. 2019 May;30(3):294-302. [PubMed: 30049244]
- 27.
- Gamil HD, Ibrahim SA, Ebrahim HM, Albalat W. Platelet-Rich Plasma Versus Tretinoin in Treatment of Striae Distensae: A Comparative Study. Dermatol Surg. 2018 May;44(5):697-704. [PubMed: 29701622]
Disclosure: Amanda Oakley declares no relevant financial relationships with ineligible companies.
Disclosure: Bhupendra Patel declares no relevant financial relationships with ineligible companies.
- Review Management of stretch marks (with a focus on striae rubrae).[J Cutan Aesthet Surg. 2017]Review Management of stretch marks (with a focus on striae rubrae).Wollina U, Goldman A. J Cutan Aesthet Surg. 2017 Jul-Sep; 10(3):124-129.
- The Objective Stretch Marks Photonumeric Assessment Scale: A New and Complete Method to Assess Striae Distensae.[Plast Reconstr Surg. 2023]The Objective Stretch Marks Photonumeric Assessment Scale: A New and Complete Method to Assess Striae Distensae.La Padula S, Hersant B, Pizza C, Chesné C, Jamin A, Ben Mosbah I, D'Andrea F, Persichetti P, Rega U, Pensato R, et al. Plast Reconstr Surg. 2023 Feb 1; 151(2):307-313. Epub 2022 Nov 9.
- Risk factors of striae gravidarum.[Int J Cosmet Sci. 2015]Risk factors of striae gravidarum.Kasielska-Trojan A, Sobczak M, Antoszewski B. Int J Cosmet Sci. 2015 Apr; 37(2):236-40. Epub 2015 Jan 12.
- Bullous striae distensae in a young patient with cardiomyopathy.[Dermatol Reports. 2023]Bullous striae distensae in a young patient with cardiomyopathy.Alowairdhi Y, Alowirdi F, Alessa D. Dermatol Reports. 2023 Dec 1; 15(4):9762. Epub 2023 Aug 9.
- Review Therapeutic targets in the management of striae distensae: A systematic review.[J Am Acad Dermatol. 2017]Review Therapeutic targets in the management of striae distensae: A systematic review.Hague A, Bayat A. J Am Acad Dermatol. 2017 Sep; 77(3):559-568.e18. Epub 2017 May 24.
- Stretch Marks - StatPearlsStretch Marks - StatPearls
- Uterine Corpus High Grade Endometrial Stromal SarcomaUterine Corpus High Grade Endometrial Stromal SarcomaMedGen
- C2239246[conceptid] (1)MedGen
- Chapter 2—Brief Interventions in Substance Abuse Treatment - Brief Interventions...Chapter 2—Brief Interventions in Substance Abuse Treatment - Brief Interventions and Brief Therapies for Substance Abuse
- MTND4P35 MT-ND4 pseudogene 35 [Homo sapiens]MTND4P35 MT-ND4 pseudogene 35 [Homo sapiens]Gene ID:107075117Gene
Your browsing activity is empty.
Activity recording is turned off.
See more...