NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Institute of Medicine (US) Committee on Military Nutrition Research; Marriott BM, editor. Food Components to Enhance Performance: An Evaluation of Potential Performance-Enhancing Food Components for Operational Rations. Washington (DC): National Academies Press (US); 1994.

Food Components to Enhance Performance: An Evaluation of Potential Performance-Enhancing Food Components for Operational Rations.
Show detailsDavid M.Penetar, 1 Una McCann,
David Thorne, Aline Schelling,
Cynthia Galinski, Helen Sing,
Maria Thomas, and Gregory Belenky
INTRODUCTION
The demands and pace of continuous military operations produce sleep fragmentation and varying degrees of sleep deprivation in soldiers. The observable effects of this sleep disruption include decreased alertness, slowed thinking, lapses in attention, decreased motivation, and a performance phenomenon called the speed-accuracy-tradeoff. Several studies have documented performance and mood changes during periods of sleep deprivation (Babkoff et al., 1989a,b; Mikulincer et al., 1989; Thorne et al., 1983). There is a decline in the performance of accuracy tasks involving attention, reasoning abilities, and reaction time. Alertness decreases, and there are changes in self-reported fatigue (increases) and vigor (decreases). Sustaining optimum soldier performance relates to the prevention or amelioration of these changes. Three categories of solutions include the following: (1) Doctrinal solutions involve the overtraining of soldiers to perform their tasks with a minimum of cognitive effort; ensuring that soldiers are cross-trained so that crew members can substitute for each other when necessary; developing and adhering to appropriate work-rest cycles, including rotating crews as the mission permits; developing efficient leadership tactics so that unnecessary demands are not placed on subordinates; and modifying equipment and systems so that the likelihood of making errors is as small as possible. (2) Behavioral solutions make sure that soldiers sleep as much as they can, whenever they can (sleep discipline), while paying attention to where and how they sleep (sleep hygiene), that is, in as comfortable a position as possible, away from the noise and bustle of other soldiers and equipment. (3) Pharmacological solutions may be warranted in special circumstances, when adherence to doctrinal and behavioral solutions is not possible or breaks down. This last category of solutions for preventing or ameliorating sleep deprivation effects is the topic of the research reported in this chapter.
The characteristics of stimulant drugs useful in specific military situations are not only effectiveness but low therapeutic toxicity and low abuse potential, d-Amphetamine was tested for its ability to reverse changes in mood, alertness, and cognitive performance after an extended period of sleep deprivation (Newhouse et al., 1989). Although effective, its restricted legal status and the psychological side effects associated with long-term use, coupled with its abuse potential, almost certainly prohibit large-scale use. Caffeine is a universally available, legal, and socially accepted and used stimulant with low toxicity and low abuse potential. It is believed to reverse the performance and mood effects seen during sleep deprivation, although it has not been tested systematically.
Caffeine has a long history of use and can be found in many common foods, drinks, and medications. Although caffeine has been the subject of pharmacological studies for several decades, the mechanism of action of its effects on the central nervous system have only recently been defined as a blockade of adenosine receptors (Choi et al., 1988; Fredholm, 1985; Snyder, 1984). Extensive reviews of caffeine (Dews, 1984; Weiss and Laties, 1962) conclude that its stimulant properties are weak in comparison with those of other drugs (e.g., amphetamine) and that its effects are modest, making detection of these effects difficult and generalizations cumbersome. Dews (1984), however, states that the following three effects are clear: (1) it has the tendency to postpone sleep; (2) it reduces the degradation of performance because of fatigue and boredom; and (3) it decreases hand steadiness. The interpretation distilled from these and other reviews is that caffeine's effects are significant primarily when performance of repetitive, nonintellectual tasks is partially degraded. What is lacking in the literature and of importance to military operations is a systematic study of caffeine's effects on sleep-deprived individuals and the assessment of caffeine's effectiveness in reversing the changes caused by sleep deprivation.
EFFECTS OF CAFFEINE ON PERFORMANCE
A common method of assessing the effects of stimulant drugs is to measure individuals' reaction times on laboratory tasks. Older reviews and several recent articles failed to find that caffeine has significant effects on the reaction times of various tasks (Bruce et al., 1986; Kuznicki and Turner, 1986; Rapoport et al., 1981; Swift and Tiplady, 1988). Recently, however, caffeine has been found to significantly decrease reaction times in auditory and visual choice tasks when it was used at doses of as little as 32 mg and as great as 600 mg (Jacobson and Edgley, 1987; Lieberman et al., 1987a,b; Roache and Griffiths, 1987). Other cognitive and psychomotor skills measured by the digit-symbol substitution task, serial subtraction of numbers, addition, symbol cancellation, card sorting, signal detection, symbol copying, and number cancellation have not been found to be affected by caffeine (Bruce et al., 1986; Childs, 1978; Ghoneim et al., 1986; Lieberman et al., 1987b; Loke et al., 1985; Roache and Griffiths, 1987; Swift and Tiplady, 1988), although Battig et al. (1984) report that 300 mg of caffeine had a positive effect on a letter cancellation test.
EFFECTS OF CAFFEINE ON MOOD AND ALERTNESS
Several questionnaires and methods have been used to assess caffeine's effects on mood. Studies have shown that doses of 200 to 250 mg of caffeine elevate mood (Lieberman et al., 1987b; Swift and Tiplady, 1988) and that these effects can last for up to 3 h. Higher doses (600 mg) can produce increases in subjects self-ratings of tension or anxiety and increases in caffeine-related physical symptoms (Roache and Griffiths, 1987). Caffeine significantly affects measures of alertness and sleepiness. Doses of 100 mg and more postpone the onset of sleep (Dews, 1982; Goldstein et al., 1965), and a dose of approximately 300 mg significantly improves daytime alertness in both partially sleep-deprived and fully rested individuals, as measured by the multiple sleep latency test (Lumley et al., 1987). Walsh et al. (1990) report that 4 mg of caffeine per kg of body weight (approximately 300 mg) significantly increases latency to sleep onset at test periods throughout the night (0100 to 0500 h). This effect was seen in light and moderate caffeine users. Multiple sleep latency test results by Rosenthal et al., (1991) showed that caffeine at doses of 75 and 150 mg increases alertness whether an individual is fully rested or partially sleep deprived. Zwyghuizen-Doorenbos et al. (1990) showed that 250 mg of caffeine improved daytime alertness for at least 3 h in moderately sleepy subjects.
PHYSIOLOGICAL EFFECTS OF CAFFEINE
The literature supports the fact that caffeine has low toxicity and produces no serious physiological side effects in adults (Rall, 1985; Stavric, 1988). Caffeine's physiological effects are dependent on whether the subject is caffeine-naive or a regular user. In caffeine-naive subjects, a 250-mg oral dose increased the systolic blood pressure by 11 mm Hg (Robertson and Curatolo, 1984). The heart rate declined during the first hour after drug administration and then increased above the baseline during the second hour. Regular users who were deprived of caffeine showed a significant increase in their systolic blood pressure (4 mm Hg), whereas regular users who were not deprived of caffeine showed no significant increases. Other researchers have found no significant changes in blood pressure or heart rate even after a 500-mg oral dose (Bruce et al., 1986; Myers 1988). Newcombe et al. (1988) showed that doses of 300 to 350 mg do not affect the prevailing cardiac rhythm or rate and do not cause clinically significant ventricular or supraventricular dysrhythmias. Specific studies concerning the cardiovascular effects of caffeine at doses above 500 mg are lacking. In several studies, however, higher doses (up to 800 mg) were administered in other contexts (Chait and Griffiths, 1983; Griffiths and Woodson, 1988). Chait and Griffiths (1983) reported that one subject experienced shakiness and an upset stomach after receiving 200 mg of caffeine, and higher doses were not tested in that subject. All other subjects tolerated doses up to 800 mg well. Other clinically relevant physiological effects include the relaxation of smooth muscles (especially the bronchii) and diuresis. Respiration rate is increased by 250-mg doses in caffeine-naive users (Curatolo and Robertson, 1983). Caffeine has the ability to increase metabolic rate, but its effect on body temperature in humans has not been reported in the literature.
In summary, studies indicate that caffeine can have significant effects on mood and performance, even at relatively low doses, in non-sleep-deprived individuals and has effects on alertness in moderately sleep-deprived individuals. The following section reports the effectiveness of caffeine in reversing the alterations seen in performance, mood, and alertness following a prolonged period of sleep deprivation.
MATERIALS AND METHODS
Subjects
Fifty normal, healthy, nonsmoking, drug-free males between the ages of 18 and 32 (mean age, 23.6 years) were recruited through advertisements in college newspapers. Subjects were within normal weight limits for their height and were moderate users of caffeine (no more than 300 mg on a daily basis). After giving free and informed written consent, subjects underwent a thorough physical and laboratory examination and completed a self-assessment questionnaire for anxiety and depression (Snaith et al., 1976). Exclusion criteria included past or present major medical or psychiatric illness, positive urine drug screen, regular tobacco use, excessive caffeine use (more than 300 mg on a daily basis), questionnaire scores above 6 (out of a possible 12) for either anxiety or depression, or atypical sleeping patterns (e.g., self-reports of difficulty in falling asleep or regular involuntary early morning awakenings). Following completion of the screening procedures, the subjects were randomly assigned to one of four drug conditions: placebo (n=12), 150 mg/70 kg (n= 13), 300 mg/70 kg (n=12), or 600 mg/70 kg (n=13).1
Procedure
Subjects arrived in the laboratory in groups of three to four each on the evening before the sleep deprivation period began. All subjects were required to refrain from caffeine and alcohol ingestion for 72 h prior to the beginning of the study. Blood and urine samples were taken to ensure that the subjects were free of caffeine and other drugs of abuse. They were trained on a computerized performance assessment battery (Thorne et al., 1985) designed to assess several cognitive functions periodically at approximately 2-h intervals throughout the sleep deprivation period (see full description below). Eleven electrodes were attached to the scalp and face by using the international 10–20 system of electrode placement. Electroencephalograms (EEGs), electrooculograms (EOGs), and submental electromyograms (EMGs) were recorded continuously by using an 8-channel Oxford Medilog ambulatory cassette recorder. Dinner was provided between training sessions. Subjects retired at 2300 h. The sleep deprivation period began upon awakening the next morning at 0700 h (day 1). They remained awake for the next 64.5 h over a 3-day period (e.g., 0700 h Tuesday morning to 2230 h Thursday night), except for brief periods during administration of the modified Multiple Sleep Latency Tests (MSLTs). Subjects were monitored continuously by the staff to prevent unintentional sleep and were provided with books, games, movies, music tapes, conversation, and occasional brief walks to occupy them between tests.
On the morning of day 3 (at 0700 h after 48 h of sleep deprivation), a Teflon catheter was inserted into a forearm vein of each subject and was maintained with a heparin lock (heparin sodium, 20 Units/ml) to facilitate repeated drawing of blood samples before and after drug administration. At 0800 h (following 49 h of sleep deprivation), subjects were administered placebo or one of three doses of caffeine, USP anhydrous (City Chemical Corporation, New York, N.Y.). Doses were either 150, 300, or 600 mg/70 kg of body weight. Caffeine was administered orally by having the subjects drink 250 ml of an artificially sweetened lemon juice drink in which the caffeine powder was dissolved. Placebo consisted solely of the sweetened lemon juice drink. All doses were given in a double-blind manner.
Performance Measures
The computerized performance assessment battery included tests of code substitution and recall, logical reasoning, sustained attention with a mental arithmetic task (referred to as the serial addition/subtraction task), match-to-sample, and choice reaction time. These tasks were administered every 2 h through the sleep deprivation period and at 1, 2, 3, 4, 6, 8, 10, and 12 h after caffeine administration.
Code Substitution and Recall. A code key that paired the digits 1 through 9 in a one-to-one correspondence with letters was presented to the subjects. Below the key, a letter appeared and subjects had to press the key of the correct number. The code key disappeared after 27 pair presentations; if needed, they could see it again by pressing 0. Fifty-four pairs were presented. Recall was assessed after the subject completed the rest of the battery tasks (about 15 min later). Subjects had to press a number in response to each of nine letter presentations; no code key was available during the recall testing.
Logical Reasoning. A task of logical reasoning ability was adapted from Baddeley (1968). The letter pair AB or BA was presented along with a statement that correctly or incorrectly described the order of the letters within the pair (e.g., “B follows A” or “A is not preceded by B”). The subject decided whether the statement was true (same) or false (different) and pressed the “S” or “D” key accordingly. The 32 possible permutations were presented once each in random order.
Sustained Attention. Sustained attention ability was assessed by the serial addition/subtraction task adapted from Pauli as used by Wever (1979, 1981). The task was a machine-paced mental arithmetic task requiring sustained attention and concentration. Two randomly selected digits and either a plus or a minus sign were displayed sequentially in the same center screen location followed by a prompt symbol: (?). The subject performed the indicated addition or subtraction and entered the least significant digit of the result. If the result was negative, the subject first added 10 to it and entered the single positive digit that was the remainder. The digits and signs were each presented for 250 ms and were separated by 200 ms, with the next trial beginning 500 ms after the response. The task ended after 50 responses and typically took 3 to 4 min.
Match-to-Sample. Subjects viewed a 6-by-6 block of squares with 36 red and green squares arranged in random order (each block contained an equal number of red and green squares). This task of immediate recall allowed subjects to view the arrangements for as long as they liked and then press a key to present two choices, one of which matched the original set of squares. They were required to pick the correct matching square. Twenty trials were presented.
Reaction Time. A choice reaction time task was used to measure reaction time. The visual-motor task required subjects to press the numbered keyboard keys corresponding to numbers presented on the screen. The digits 0 through 9 appeared one at a time in the center of the screen. The stimulus remained on until a response was made. Fifty numbers were presented. This task was designed to fulfill the requirements of a standard reaction time task while closely resembling the physical requirements of the serial addition/subtraction task.
Mood Measures
Profile of Mood States. The Profile of Mood States (POMS) (McNair et al., 1981) is a 65-item adjective checklist that measures current mood states along six subscales: tension-anxiety, anger-hostility, depression-dejection, vigor-activity, fatigue-inertia, and confusion-bewilderment. Subjects rated themselves on each adjective from 1 (not at all) to 5 (extremely).
Visual Analog Scales (VAS). Subjects rated themselves with a mark along a line 100-mm in length. The lines were labeled “alert/able to concentrate,” “anxious,” “energetic,” “feel confident,” “irritable,” “jittery/nervous,” “sleepy,” and “talkative.” Additional ratings were obtained on four possible effects of sleep deprivation and caffeine administration. They were heart pounding, headache, sweaty, and upset stomach. The lines were labeled “not at all” on the left end and “extremely” on the right end.
The POMS and VAS were completed five times during the sleep deprivation period prior to drug administration at 0900 and 2000 h on days 1 and 2 and at 0600 h on the morning of day 3 (2 h before caffeine administration). POMS and VAS ratings were taken at 1, 2, 4, 8, and 12 h after drug administration.
Alertness Measures
Multiple Sleep Latency Tests. Each modified multiple sleep latency test (MSLT) was conducted by having the subjects lie in bed in a darkened, sound-attenuated room with their eyes closed. They were instructed to relax and allow themselves to fall asleep. EEGs, EOGs, and EMGs were displayed on a Grass Electroencephalograph (model 8–10D) for on-line scoring of awake versus sleep during the MSLT. An experimenter awakened a subject after 30 s of stage 2 sleep or the onset of rapid eye movement (REM). The test was terminated at 20 min if sleep had not occurred. MSLTs were conducted at 0930, 1130, 1430, 1630, 1830, and 2030 h on days 1 and 2 of the sleep deprivation period. MSLTs were conducted eight times on day 3 (at 1.5, 2.5, 3.5, 4.5, 6.5, 8.5, 10.5, and 12.5 h after drug administration).
Stanford Sleepiness Scale. For the Stanford Sleepiness Scale (SSS) (Hoddes et al., 1973), subjects selected one of seven statements that best described their current state of alertness, ranging from 1 (feeling active and vital; alert; wide awake) to 7 (almost in reverie; sleep onset soon; losing struggle to remain awake). The SSS was completed approximately every 2 h throughout the sleep deprivation period (at 0900, 1100, 1400, 1600, 1800, 2000, 2200, 2400, 0200, 0400, and 0600 h on days 1 and 2). The SSS was completed eight times on day 3 (at 1, 2, 3, 4, 6, 8, 10, and 12 h after drug administration).
Vital Signs
Measurements of blood pressure, heart rate, and oral temperature were taken at least every 2 h throughout the sleep deprivation period. After caffeine administration, measurements were taken at 15, 30, 60, 90, 120, 150, 180, and 210 min and then hourly until 13 h after drug administration.
Catecholamine and Caffeine Assays
Blood samples were collected prior to and at 15, 30, 60, and 90 min, and 2, 2.5, 3, 4, 6, 8, 10, and 12 h following drug administration. Results are reported elsewhere (Eddington et al., 1993; and Penetar et al., 1993).
Statistical Analysis
Separate two-factor repeated measures analysis of variance by using the General Linear Model (SAS Institute, Cary, N.C.) were performed for each dependent variable and POMS subscale. The two factors were group or dose and time. First, each dependent variable was analyzed for group differences and effects of the sleep deprivation period prior to the drug administration by using all measurements made prior to drug administration. Second, each dependent variable was analyzed for the effects of drug and time after drug administration by using the last value obtained prior to drug administration and all values obtained after drug administration. Statistical results thus reported for the main effects of drug dose, the main effects of time, and an interaction between these main effects. A level of P<0.05 was accepted as significant, with the Greenhouse-Geisser adjustment criterion used in evaluating the main effects of time and the interaction between the main effects. Significant main effects were further evaluated by the Newman-Keuls Multiple Range Test.
RESULTS
Performance Tests
For each of the tasks, three measures of performance were analyzed: accuracy (percent correct), speed (responses per unit of time), and throughput (number of correct responses per unit of time). The throughput measure takes both accuracy and speed of performance into account and was subjected to statistical testing. Significant effects of caffeine were observed on the throughput measure for the choice reaction time, serial addition/subtraction, and logical reasoning tasks (Figure 20–1 and Table 20–1).
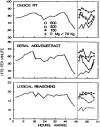
FIGURE 20–1
Mean throughput measures for three performance tasks: choice reaction time (Choice RT), serial addition/subtraction, and logical reasoning. Predrug means include data for all subjects; there were no differences between the groups prior to drug administration. (more...)
TABLE 20–1
Performance, Mood, and Physiology Analysis of Variance Summary.
Performance on the choice reaction time task for 8 h after drug administration in subjects who received the 600-mg dose was significantly different from that in subjects who received the placebo. The 150-mg dose improved performance for 4 h. For subjects receiving the 300-mg dose, performance was not significantly different from that for subjects receiving the placebo at any point following administration. For the serial addition/subtraction task, all doses of caffeine significantly improved performance for 3 h. For subjects receiving the 600-mg dose, performance remained significantly better than that for subjects receiving placebo for 10 h after drug administration, with no significant differences observed among the dose groups at the final 12-h testing period. Performance on the logical reasoning task by subjects receiving the two highest doses of caffeine was significantly better than by subjects receiving placebo for the entire 12-h period. In addition, caffeine restored performance to the levels obtained after rest during this interval. Performance after administration of the 150-mg dose was significantly different from that after administration of placebo for 6 h after drug administration.
Mood Measures
The effects of sleep deprivation on mood, as measured by the POMS and VAS, are reported in more detail elsewhere (Penetar et al., 1993). Briefly, the scores of all six subscales of the POMS changed significantly as a result of the sleep deprivation. Tension [F(5,230)=12.3, P<0.001], anger [F(5,230)= 7.42, P<0.001], depression [F(5,230)=16.4, P<0.001], fatigue [F(5,230) =97.9, P<0.001], and confusion [F(5,230)=57.4, P<0.001] increased, whereas vigor decreased [F(5,230)=87.3, P<0.001]. Similarly, ratings on the VAS showed the effects of sleep deprivation. The main effects for the time before drug administration showed that alertness [F(4,184)=103.39, P< 0.001], energy levels [F(4,184)=109.2, P<0.001], confidence [F(4,184)= 48.96, P<0.001], and talkativeness [F(4,184)=23.46, P<0.001] declined, whereas irritability [F(4,184)=20.29, P<0.001] and sleepiness [F(4,184)= 138.11, P<0.001] increased.
Following caffeine administration, significant increases in the POMS vigor subscale and significant decreases in the POMS subscales of fatigue and confusion were observed (Table 20–1). Vigor ratings for all three dose groups were significantly different from those for the placebo group for 2 h after caffeine adminstration. Vigor ratings for the 600-mg dose group were 97 percent of those for subjects in the rested condition 1 h after caffeine administration and remained at 84 percent of those for subjects in the rested condition at the 2-h measurement. Conversely, fatigue ratings for all three caffeine dose groups decreased significantly for 2 h following caffeine administration. Confusion ratings in the 150-mg dose group were significantly decreased in comparison with those in the placebo group 2 h after caffeine administration.
Caffeine reversed the sleep deprivation effects reported in subjective ratings of alertness for 2 h, energy levels for 12 h, confidence for 2 h, sleepiness for 12 h, and talkativeness for 2 h following drug administration. Caffeine significantly increased self-rated anxiety for 2 h, and jitteriness or nervousness for 12 h following drug administration. Ratings of heart pounding, headache, sweatiness, and upset stomach were unaffected by caffeine.
Alertness Measures
Multiple Sleep Latency Tests
For the rested condition (day 1), mean sleep latency periods were between 16.3 and 19.9 min. Sleep deprivation significantly [F(11,506)=202.39, P<0.001] decreased latency periods to a range of 5.6 to 7 min on day 2. Significant dose [F(3,46)=5.18, P<0.005], time [F(7,322)=5.61, P<0.001] and time×dose interactions [F(21,322)=1.99, P<0.05] were observed following caffeine administration. Latency periods for the highest dose (600 mg/70 kg) group increased significantly above placebo latency periods for the placebo group (10.2 min versus 5.0 min) (Figure 20–2) and remained at this level for 4.5 h after drug administration. The dose of 300 mg/70 kg appeared to have effects intermediate between those of the highest dose tested and the placebo; however, these effects were not significantly different from those for the other dose groups.

FIGURE 20–2
Latency to stage 2 sleep following caffeine administration. *, results for the 600-mg dose group were significantly different from those for the placebo group (P<0.05).
Stanford Sleepiness Scale
Average values of the Stanford Sleepiness Scale increased gradually from 1.6 at 0900 h on day 1 (subjects rested) to a maximum average of 4.8 at 0600 h on the morning of day 3, indicating a significant effect of sleep deprivation (Table 20–2). Caffeine's effects were significant for 2 h after drug administration and were not dose-related (i.e., all doses were equally effective).
TABLE 20–2
Stanford Sleepiness Scale Scores.
Vital Signs
Diastolic blood pressure and oral temperature were significantly affected by caffeine administration (Table 20–1 and Figure 20–3). At 1 h after admini stration, both the 300- and the 600-mg doses significantly increased diastolic blood pressure in comparison with the placebo; there were no significant differences at other time points. The 600-mg dose of caffeine significantly increased oral temperature in comparison with placebo at several measurement times after administration: 2, 2.5, 3, 4, 6, 8, and 12 h. Neither systolic blood pressure nor pulse was significantly affected.
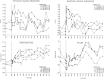
FIGURE 20–3
Time course of caffeine effects on four vital signs. Caffeine was observed to have significant effects on diastolic blood pressure (at 1 h) and oral temperature from 2 to 12 h after drug administration. See text for details.
DISCUSSION
The study described here indicates that caffeine is effective in reversing the performance degradations and the alterations in mood and alertness produced by periods of prolonged sleep deprivation. The results indicate that these beneficial effects can be long-lasting and not at the expense of serious mood or physiological side effects.
Sleep deprivation degrades cognitive performance. The effects of caffeine on performance in non-sleep-deprived volunteers have been well documented, even at the low dose levels commonly found in food and drink products (see Lieberman [1992] for a review). The study described here extends the usefulness of caffeine, showing that large doses (up to 600 mg) are effective in improving a variety of cognitive performances in sleep-deprived individuals, and outlines the time course of its effects in these individuals. The tasks used in the present study were chosen to sample a variety of cognitive abilities with varying mental demands. Choice reaction time requires little thinking but does require great accuracy and speed. The serial addition/subtraction task is a machine-paced task but has a greater mental component, whereas the logical reasoning task is self-paced and requires the greatest amount of thought for accurate responses. Caffeine produced improved performances of all three tasks, with performance returning to those of rested subjects for up to 12 h after caffeine administration. Caffeine was not observed to affect recall or code substitution tasks. In toto, these results are in concert with those presented previously (Lieberman, 1992; Roache and Griffiths, 1987) and document for the first time the relatively long-lasting effects of this drug on cognitive performance. The study described here shows that caffeine compares favorably with amphetamine in reversing the effects of sleep deprivation on cognitive performance. Using an identical sleep deprivation paradigm, Newhouse et al. (1989) showed that 20 mg of amphetamine is required to produce sustained performance improvements.
Sleep deprivation also alters mood and degrades alertness. The present study documents the fact that caffeine can have significant beneficial effects in reversing these mood changes; sleepiness and confusion declined, whereas increases in energy and confidence levels were reported. Although there were increased ratings of anxiety and jitteriness or nervousness, these effects were not severe and did not elicit complaints from the subjects. Depending on the measure, alertness, which was severely degraded by 49 h of sleep deprivation, was improved for 2 to 4.5 h, whereas self-ratings of sleepiness in groups treated with caffeine remained significantly lower than those in the placebo group for 12 h. In this regard, caffeine was not as effective as amphetamine. The alertness of amphetamine treated subjects (20 mg), as measured by sleep latency tests, was nearly restored to the levels of rested subjects for 7 h (Newhouse et al., 1989). Caffeine's effects on alertness are therefore less potent and shorter acting than amphetamine's.
Caffeine's effects on physiological measures are important for assessing its usefulness as a stimulant. The study described here shows that relatively high doses of caffeine are well tolerated by sleep-deprived individuals and that its effects are similar to those found in other studies in non-sleep-deprived subjects given lower doses than those used in the present study (Myers, 1988; Newcombe et al., 1988). Additionally, there were no changes in self-reports of other side-effects (heart pounding, headache, sweatiness, upset stomach). Of note was caffeine's observed effect on oral temperature. Oral temperature normally rises during the day, from a low in the early morning hours to a peak in the early evening hours. The subjects in the present study showed this typical response. Caffeine increased temperatures above the normal rise throughout the observation period, again revealing an important aspect of its effects and duration of action. The significance of this effect awaits further experimentation, although this type of effect has been observed previously with another stimulant, d-amphetamine (Newhouse et al., 1989).
RECOMMENDATIONS
- The results of the present study would seem to indicate that caffeine can be an ideal stimulant for use during military operations when performance declines secondary to sleep disruption and sleep fragmentation. Doses of 600 mg are needed to reverse severely degraded performance as a result of long periods of sleep deprivation. Presumably, lower doses (200–400 mg) would be effective in ameliorating the changes caused by shorter periods of deprivation.
- Given the legal and social acceptance of caffeine, and its low abuse potential, caffeine tablets (200 mg/tablet) should be included in food rations.
- Use of caffeine should be restricted to special situations when sleep has been unusually disrupted and for the benefit of temporarily (10–12 h) restoring alertness and sustaining performance during critical periods of military operations.
- Finally, although caffeine can temporarily sustain performance during continuous operations, it should be emphasized that no drug can substitute for adequate sleep.
ACKNOWLEDGMENTS
The authors thank the staff of the Behavioral Biology, in particular Sharon Balwinski and Kevin Peyton, for assistance in the conduct of the experiment described here. Investigators adhered to AR 70–25 and USAMRDC Reg 70–25 on the use of volunteers in research. Use of trade names does not constitute endorsement of product. The views of the authors do not purport to reflect the position of the Department of the Army or the Department of Defense.
REFERENCES
- AR 70–25 1989. See U.S. Department of the Army. 1989.
- Babkoff, H., M.Mikulincer, T.Caspy, R.L.Carasso, and H.C.Sing 1989. a The implications of sleep loss for circadian performance accuracy. Work Stress 3:3–14.
- Babkoff, H., H.C.Sing, D.R.Thorne, S.G.Genser, and F.W.Hegge 1989. b Perceptual distortions and hallucinations reported during the course of sleep deprivation. Perceptual Motor Skills 68:787–798. [PubMed: 2748294]
- Baddeley, A.D. 1968. A 3-minute test based on grammatical transformations. Psychonomic Sci. 10:341–342.
- Battig, K., R.Buzzi, J.R.Martin, and J.M.Feierabend 1984. The effects of caffeine on physiological functions and mental performance. Experentia 40:1218–1223. [PubMed: 6500006]
- Bruce, M., N.Scott, M.Lader, and V.Marks 1986. The psychopharmacological and electrophysiological effects of single doses of caffeine in healthy human subjects. Br. J. Clin. Pharmacol. 22:81–87. [PMC free article: PMC1401080] [PubMed: 3741730]
- Chait, L.D., and R.R.Griffiths 1983. Effects of caffeine on cigarette smoking and subjective response. Clin. Pharmacol. Ther. 34:612–622. [PubMed: 6627821]
- Childs, J.M. 1978. Caffeine consumption and target scanning performance. Hum. Factors 20:91–96. [PubMed: 640697]
- Choi, O.H., M.T.Shamin, W.L.Padgett, and J.W.Daly 1988. Caffeine and theophylline analogues: Correlation of behavioral effects with activity as adenosine receptor antagonists and as phosphodiesterase inhibitors. Life Sci. 43:387–398. [PubMed: 2456442]
- Curatolo, P.W., and D.Robertson 1983. The health consequences of caffeine. Ann. Intern. Med. 98:641–653. [PubMed: 6342491]
- 1984. Behavioral effects of caffeine. Pp. 86–103 in Caffeine, P.B Dews, editor. , ed. New York: Springer-Verlag.
- Eddington, N.D., S.I.Lugo, G.Kamimori, E.Fadiran, D.Penetar 1993. The influence of caffeine administration on in-vivo catecholamine pharmacodynamics in sleep deprived volunteers. Pharmacol. Res., 10:S-360.
- Fredholm, B.B. 1985. On the mechanism of action of theophylline and caffeine. Acta Med. Scand. 217:149–153. [PubMed: 2986418]
- Ghoneim, M.M., J.V.Hinrichs, C.K.Chiang, and W.H.Loke 1986. Pharmacokinetic and pharmacodynamic interactions between caffeine and diazepam. J. Clin. Psychopharmacol. 6:75–80. [PubMed: 3517081]
- Goldstein, A., R.Warren, and S.Kaizer 1965. Psychotropic effects of caffeine in man. I. Individual differences in sensitivity to caffeine-induced wakefulness. J. Clin. Pharmacol. Exp. Therapeutics 149:156–169. [PubMed: 14334284]
- Griffiths, R.R., and P.P.Woodson 1988. Reinforcing effects of caffeine in humans. J. Pharmacol. Exp. Ther. 246:21–29. [PubMed: 3392652]
- Hoddes, E., V.Zarcone, H.Smythe, R.Phillips, and W.Dement 1973. Quantification of sleepiness: A new approach. Psychophysiology 10:431–436. [PubMed: 4719486]
- Jacobson, B.H., and B.M.Edgley 1987. Effects of caffeine on simple reaction time and movement time. Aviat. Space Environ. Med. 58:1153–1156. [PubMed: 3426488]
- Kuznicki, J.T., and L.S.Turner 1986. The effects of caffeine on caffeine users and non-users. Physiol. Behav. 37:397–408. [PubMed: 3749299]
- Lieberman, H.R. 1992. Caffeine. Pp. 49–72 in Factors Affecting Human Performance, Vol. 2: The Physical Environment, D.Jones, editor; and A.Smith, editor. , eds. London: Academic Press.
- Lieberman, H.R., R.J.Wurtman, G.G.Emde, and I.L.G.Coviella 1987. a The effects of caffeine and aspirin on mood and performance. J. Clin. Pharmacol. 7:315–320. [PubMed: 3680601]
- 1987. b The effects of low doses of caffeine on human performance and mood. Psychopharmacology 92:308–312. [PubMed: 3114783]
- Loke, W.H., J.V.Hinrichs, and M.M.Ghonheim 1985. Caffeine and diazepam: Separate and combined effects on mood, memory, psychomotor performance. Psychopharmacology 87:344–350. [PubMed: 3936091]
- Lumley, M., T.Roehrs, D.Asker, F.Zorick, and T.Roth 1987. Ethanol and caffeine effects on daytime sleepiness/alertness. Sleep 10:306–312. [PubMed: 3659729]
- McNair, D.M., M.Lorr, and L.F.Droppleman 1981. Profile of Mood States. San Diego, Calif: Educational and Industrial Testing Service.
- Mikulincer, M., H.Babkoff, T.Caspy, and H.Sing 1989. The effects of 72 hours of sleep loss on psychological variables. Br. J. Psychol. 80:145–162. [PubMed: 2736337]
- Myers, M.G. 1988. Effects of caffeine on blood pressure. Arch. Inter. Med. 148:1189–1193. [PubMed: 3284502]
- Newcombe, P.F., K.W.Renton, P.M.Rautaharju, C.A.Spencer, and T.J.Montague 1988. High-dose caffeine and cardiac rate and rhythm in normal subjects. Chest 94:90–94. [PubMed: 2454782]
- Newhouse, P.A., G.Belenky, M.Thomas, D.Thorne, H.C.Sing, and J.Fertig 1989. The effects of d-amphetamine on arousal, cognition, and mood after prolonged total sleep deprivation. Neuropsychopharmacology 2:153–164. [PubMed: 2742729]
- Penetar D., U.McCann, D.Thorne, G.Kaminori, G.Galinski, H.Sing, M.Thomas, G.Belenky 1993. Caffeine reversal of sleep deprivation effects on alertness and mood. Psychpharmacology, 112:359–365. [PubMed: 7871042]
- Rall, T.W. 1985. Central nervous system stimulants: The methylxanthines. Pp. 589–603 in The Pharmacological Basis of Therapeutics, A.G.Gilman, editor; , L.S.Goodman, editor; , T.W.Rall, editor; , and F.Murad, editor. , eds. New York: Macmillan Publishing Company.
- Rapoport, J.L., M.Jensvold, R.Elkins, M.S.Buchsbaum, H.Weingartner, C.Ludlow, T.P. Zahn, C.J.Berg, and A.H.Neims 1981. Behavioral and cognitive effects of caffeine in boys and adult males. J. Nervous Mental Dis. 169:726–732. [PubMed: 7299385]
- Roache, J.D., and R.R.Griffiths 1987. Interactions of diazepam and caffeine: Behavioral and subjective dose in humans. Pharmacol. Biochem. Behav. 26:801–812. [PubMed: 3602037]
- Robertson, D., and P.W.Curatolo 1984. The cardiovascular effects of caffeine. Pp. 77–85 in Caffeine, P.B.Dews, editor. , ed. New York: Springer-Verlag.
- Rosenthal, L., T.Roehrs, A.E.Zwyghuizen-Doorenbos, D.Plath, and T.Roth 1991. Alerting effects of caffeine after normal and restricted sleep. Neuropsychopharmacology, 4:103–108. [PubMed: 2025377]
- Snaith, R.P., G.W.K. Bridge, and M.Hamilton 1976. The Leeds scales for the self-assessment of anxiety and depression. Br. J. Psychiatry 128:156–165. [PubMed: 942576]
- Snyder, S.H. 1984. Adenosine as a mediator of the behavioral effects of xanthines. Pp. 129–141 in Caffeine, P.B.Dews, editor. , ed. New York: Springer-Verlag.
- Stavric, B. 1988. Methylxanthines: Toxicity to humans. 2. Caffeine. Fed. Chem. Toxicol. 26:645–662. [PubMed: 3053369]
- Swift, C.G., and B.Tiplady 1988. The effects of age on the response to caffeine. Psychopharmacology 94:29–31. [PubMed: 3126525]
- Thorne, D.R., S.G.Genser, H.C.Sing, and F.W.Hegge 1983. Plumbing human performance limits during 72 hours of high task load. Pp. 17–40 in Proceedings of the 24th DRG Seminar on The Human as a Limiting Element in Military Systems. Toronto: Defence and Civil Institute of Environmental Medicine.
- 1985. The Walter Reed performance assessment battery. Neurobehav. Toxicol. Teratol. 7:415–18. [PubMed: 3840579]
- U.S. Department of the Army. 1989. Army Regulation 70–25. “Use of Volunters As Subjects of Research”. 25 October. Washington, D.C..
- Walsh, J.K., M.J.Muehlbach, T.Humm, Q.S.Dickens, J.L.Sugerman, and P.K.Schweitzer 1990. Effect of caffeine on physiological sleep tendency and ability to sustain wakefulness at night. Psychopharmacology 101:271–273. [PubMed: 2349369]
- Weiss, B., and V.G.Laties 1962. Enhancement of human performance by caffeine and the amphetamines. Pharmacol. Rev. 14:1–36. [PubMed: 14005855]
- Wever, R. 1979. The Circadian System of Man. Berlin: Springer-Verlag.
- 1981. On varying work-sleep schedules: The biological perspective. Pp. 51–86 in The Twenty-four Hour Workday: Proceedings of a Symposium on Variations in Work-Sleep Schedules, L.C.Johnson, editor; , D.I.Tepas, editor; , W.P.Colquhoun, editor; , and M.J.Cilligan, editor. , eds. DHHS (NIOSH) Publication #81–127. Cincinnati: Department of Health and Human Services.
- Zwyghuizen-Doorenbos A, T.A.Roehrs, L.Lipschutz, V.Timms, and T.Roth 1990. Effects of caffeine on alertness. Psychopharmacology 100:36–39. [PubMed: 2296626]
DISCUSSION
HARRIS LIEBERMAN: We have some unpublished data from a couple of studies in which we did find significant effects of caffeine on mood swing. We did not use doses as low as 32 milligrams but used doses of 64 and 128 milligrams of caffeine. Effects on performance by doses lower than those are hard to detect, but over the long run, over a series of studies, my feeling is that there really are effects with low dosages, and those are the doses that we typically take in our background.
JOEL GRINKER: I was just curious whether in any of the caffeine studies or in any of the other supplement studies age has been looked at systematically as a factor. I have two thoughts, one, that in fact it might potentiate the ability of older individuals or that in fact it has less effectiveness, and I wonder if you have any comments.
DAVID PENETAR: I have here one study that related to age. Typically, these studies were done, with young, healthy males.
HARRIS LIEBERMAN: We did look at the age parameter in one of our caffeine studies, but we did not see any significant differences as a function of age or gender.
JOHN VANDERVEEN: I was curious as to the uniformity of the synthesis associated with sleep. Do you find much variation that would indicate that shortness of sleep time versus onset of sleep, etc. Are they uniform in your subjects or are they highly individualistic?
DAVID PENETAR: In terms of a rested individual?
JOHN VANDERVEEN: Yes.
DAVID PENETAR: What we do is we bring them into the study the night before and give them 9 hours of time in bed before we start the study, so at that time they are all pretty consistent in the amount of sleep that they have had.
JOHN VANDERVEEN: No, I meant the effects of caffeine on their subsequent sleep. Do you find a uniform effect in terms of delay of sleep or shortness of length of sleep, etc?
DAVID PENETAR: We did not specifically look at that because by the time they went to bed it was over 12 hours after they had received caffeine.
WILLIAM WATERS: A couple of questions. One pertaining to the onset of parameters. Did you have a look at whether or not you have any data or whether or not sleep can be induced prior to that?
DAVID PENETAR: No, I do not have any data on that. I am not sure that is reported.
WILLIAM WATERS: It could be that what you had was a referral of something that might allow it to occur. The other thing was, under the influence of caffeine, did you notice any change in the number, the length, stage one, and arousals?
DAVID PENETAR: Again, by the time our subjects went to bed, it was over 12 hours after they had received the caffeine, and we did not see any changes; there were no differences between the groups. We did monitor them. We recorded them through their sleep, and we saw no differences in sleep architecture, time of sleep, time to bed, or sleep efficiency; we saw no differences for 12 hours.
JOHANNA DWYER: I worked with a neurologist who was interested by some observations years ago, when they did a lot more electroconvulsive shock than they do now. Apparently, they used to prime the patients with caffeine, and by doing this, they could use a lower level of shock and still get the same effect. The reason I bring it up here is not because I hope anyone here is heavily into this, but rather, are there other changes in the electroencephalograms in terms of caffeine's effects that may be in addition to what we have been talking about?
DAVID PENETAR: No, I do not know.
ALLISON YATES: Just one thing. I noticed in some of the graphs that it almost looked as if at 600 milligrams the subjects might have had even a little bit better performance than they had initially in their first 24 hours. This result is important in considering enhancement of performance with normal subjects.
DAVID PENETAR: Harris, your subjects could not sleep at night.
HARRIS LIEBERMAN: Yes, two slides that you showed with my studies, the vigilance and reaction times, were for subjects who had stayed up all night the night before and who were back in the morning after the administration. Their performances were similar to those with placebos under the same conditions. I consider that to be above normal, although since caffeine is such a common component of the diet, it is hard to untangle it all.
ALLISON YATES: That is why I was wondering yesterday what the baseline levels were.
HARRIS LIEBERMAN: We typically include that as a parameter in our studies and look to see whether there are differences between moderate, low, and heavy caffeine users in their responsiveness, and in the low and moderate range there is not much difference. When you get to the real high users, you see big differences in responsiveness. That depends on the timing of administration, whether they are in a deprivation stage, or whether they are already on a lot caffeine.
ROBERT NESHEIM: Were you defining it high?
HARRIS LIEBERMAN: Average caffeine consumption is about 200 milligrams per day, which is maybe three cups of not very strong coffee. I define high for the purpose of categorizing subjects as above 400 or 500 milligrams per day.
DAVID SCHNAKENBERG: Just a couple of observations. We used to always think that members of the Army must be heavy coffee drinkers because you get that perception, but looking out in field studies where soldiers are eating rations, we found out that even though you gave a meal ready-to-eat, 90 percent of the coffee packets were returned unused. The rest of the 10 percent probably went mostly to the senior sergeants, who had a chance to stay by the talking place and make some coffee for themselves. So young soldiers in the field today are not heavy coffee drinkers. I am sure they drink plenty of caffeine if they have carbonated beverages. But most of the time carbonated beverages are not available to them in the field, although maybe in Desert Storm cans of Coke manged to get inside of the tanks anyway.
My question is, has anybody done sleep studies on evaluating caffeine using the vehicle of delivering the caffeine in the form of a cola or in the form of a coffee beverage itself?
DAVID PENETAR: A number of studies look at coffee drinking when they give caffeine. In fact, in some of the studies reported here, they took decaffeinated coffee and added caffeine to it, and the subjects drank it that way. In other studies it was either caffeine pills or caffeine powder dissolved in some drink.
DAVID SCHNAKENBERG: Can subjects get a comparable effect with the caffeinated coffee versus decaffeinated coffee? For instance, I am sure your subjects knew when they were receiving a placebo.
DAVID PENETAR: Ours was powdered caffeine dissolved in a lemon juice drink, and the lemon juice drink was very bitter. As you know, caffeine powders are very bitter, so they could not tell what they were drinking other than lemon juice drink. If they talked among themselves, they just said “oh, it is not very good, it was bitter.”
WILLIAM BEISEL: So many of the emergency rations and so on seem to be candy bars with chocolate flavoring. How much of that is caffeine?
DAVID PENETAR: Milk chocolate has about 7 milligrams per ounce, whereas bakery chocolate or unsweetened chocolate has about 35 milligrams per ounce. They figure that, for example, a Hershey's candy bar has 25 to 35 milligrams per ounce, so it is not a lot, and it is less than most sodas.
Footnotes
- 1
David M.Penetar, Walter Reed Army Institute of Research, Washington, D.C. 20307–5100
- 1
Subjects were paid for their participation. The investigators adhered to AR 70–25 (U.S. Department of the Army, 1989) and U.S. Army Medical Research and Development Command Reg 70–25 (1989), on the use of volunteers in research.
- Effects of Caffeine on Cognitive Performance, Mood, and Alertness in Sleep-Depri...Effects of Caffeine on Cognitive Performance, Mood, and Alertness in Sleep-Deprived Humans - Food Components to Enhance Performance
- MIR299 microRNA 299 [Homo sapiens]MIR299 microRNA 299 [Homo sapiens]Gene ID:407023Gene
- TMEM192 transmembrane protein 192 [Homo sapiens]TMEM192 transmembrane protein 192 [Homo sapiens]Gene ID:201931Gene
- CSRNP2 cysteine and serine rich nuclear protein 2 [Homo sapiens]CSRNP2 cysteine and serine rich nuclear protein 2 [Homo sapiens]Gene ID:81566Gene
- 1700030K09Rik RIKEN cDNA 1700030K09 gene [Mus musculus]1700030K09Rik RIKEN cDNA 1700030K09 gene [Mus musculus]Gene ID:72254Gene
Your browsing activity is empty.
Activity recording is turned off.
See more...