| NCBI National Center for Biotechnology Information |  |
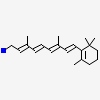
4RUU: Crystal structure of the Q108K:K40L mutant of human Cellular Retinol Binding ProteinII in complex with All-trans-Retinal after 24 hour incubation at 1.4 Angstrom Resolution |
Citing MMDB
 Madej T, Lanczycki CJ, Zhang D, Thiessen PA, Geer RC, Marchler-Bauer A, Bryant SH. " MMDB and VAST+: tracking structural similarities between macromolecular complexes. Nucleic Acids Res. 2014 Jan; 42(Database issue):D297-303
Madej T, Lanczycki CJ, Zhang D, Thiessen PA, Geer RC, Marchler-Bauer A, Bryant SH. " MMDB and VAST+: tracking structural similarities between macromolecular complexes. Nucleic Acids Res. 2014 Jan; 42(Database issue):D297-303