Polymicrobial diseases are difficult to reproduce and to study. In vivo and in vitro models are often used to study specific microbial interactions or parameters associated with infection and disease. In vivo models can determine (i) the predisposing conditions and situations leading to polymicrobial disease, (ii) the complex interactions among microorganisms in these diseases, (iii) the complex interactions between microorganisms and the host, and (iv) the mechanisms of pathogenesis leading to the clinical signs and lesions characteristic of the disease. Many of these in vivo models are discussed throughout the book and therefore are outside of the scope of this chapter. Here we focus on the in vitro techniques that have identified mechanisms of interspecies and intergeneric cooperation among microorganisms.
In Vitro Models
A traditional approach taken through the years in the study of almost all aspects of microbial physiology, metabolism, expression of virulence factors, and interaction with immune cells has been the batch-culture technique. Multiple ways in which bacteria can be grown in this manner exist, including test tubes, flasks, large vessels, etc. This has been traditional mainly because of the ease of preparing bacterial cultures and the ability to do so in almost any laboratory. Sophisticated equipment is not needed to prepare a batch culture of an organism. Although it is easy to obtain a bacterial suspension in this manner, however, numerous disadvantages of working with these cells exist. First and foremost, bacteria under their normal conditions in nature, whether in infections in a susceptible host or involved in the development of plaque in the oral cavity, simply do not grow at the rate they do in batch culture. Studies have shown, in fact, that bacteria freshly isolated from infections do not express the same profile of cell wall proteins as they do in batch culture. Moreover, many examples are available of bacteria that phenotypically adapt to antimicrobials in batch culture, or lose resistance upon subculturing. It is a well-known phenomenon that bacteria lose virulence and many other characteristics if they are subjected to extensive batch subculture.
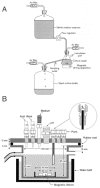
Most batch cultures involve bacteria cultivated in a continuous culture (49). Here, planktonic cultures in a fermentor are fed with a nutrient solution to maintain a bacterial population in the exponential or log phase of growth (Fig. 1A). The continuous culture reaches "balanced growth" in which the levels of bacteria, bacterial products, media components, and waste products are constant (5). This condition is referred to as "steady-state" growth (49). The culture volume and the cell concentration are both kept constant by allowing fresh, sterile medium to enter the culture vessel at the same rate that "spent" medium, containing cells, is removed from the growing culture. Under these conditions, the rate at which new cells are produced in the culture vessel is exactly balanced by the rate at which cells are being lost through the overflow from the culture vessel (49).
One type of system that is widely used for continuous cultivation is the chemostat (49). This system depends on the fact that the concentration of an essential nutrient within the culture vessel controls the growth rate of the cells. In general, one nutrient is limited to an amount that restricts growth, and the culture is removed at the same rate as nutrients are added. Steady-state bacterial growth often forms films called biofilms on various surfaces placed in these chambers (Fig. 1B). These include acrylic, glass, or hydroxylapatite attached to titanium wire or a glass rod and immersed below the surface of the culture medium (5). Hydroxyapatite surfaces are often used because they reflect the chemical composition of enamel surfaces. Biofilms are microbial stacks or columns of microcolonies often embedded in extracellular polymeric matrix (13). The biofilm is permeable and contains pores or channels in interconnected mushroom or tulip shapes (30).
Biofilms are now thought to be the natural, environmental form of bacterial growth (2, 11, 14) as well as the form often seen in certain diseases (13) including periodontal disease (chapter 8), otitis media (chapter 14), and cystic fibrosis (53). Next we present two of the most common in vitro methods used to investigate microbial interactions: continuous-culture chemostat systems and flowcells.
Continuous-Culture Chemostat Systems
An approach that provides a spectrum of exciting possibilities for studying bacteria under conditions that more closely resemble the way they grow naturally is continuous culture (Table 1). The foundation of chemostat theory was originally described by Monod (44) in France and Novick and Szilard (45) in the United States, with the mathematical nuances refined by Herbert (21) in the United Kingdom. A description of the mathematical derivations can be found elsewhere (24). In summary, when steady-state conditions are reached in the chemostat, the dilution rate (D) is equal to the specific growth rate (μ). Therefore, one can easily calculate the mean doubling time or generation time of the microorganism in culture. The key result of such a system is that bacterial growth can be manipulated to achieve slow growth rates, which more closely resemble those found in nature. By doing so, studies have shown that bacteria grown under these conditions express cell wall proteins and respond to environmental stimuli much like cells in vivo. One can begin to see how studying bacteria grown under these conditions can be powerful indeed. Moreover, a powerful approach using this methodology has been the study of mixed cultures. Many interactions have been studied, from how communities of organisms interact and develop through time to the effect of environmental insults on the homeostasis of bacterial communities (3–9, 24, 26, 27, 33–40, 51, 52, 54, 57). Excellent examples of the use of continuous-culture techniques have been in the field of oral microbiology. Marsh et al. (35) studied the influence of growth rate and nutrient limitation on the microbial composition of a community of organisms in a chemostat. Samples of human plaque were used to inoculate the chemostats, grown under glucose-excess or glucose-limiting conditions. A complex mixture of end products was analyzed, along with the cell yields, pH, and changes in the community as the composition changed over time. The results of this study can be found elsewhere; the key point to be made here is that it was determined that the continuous-culture system could be an invaluable tool to study the ecology of dental plaque. Subsequent studies by this research group focused on many aspects of plaque-community development (9, 10, 19, 39–42).
Table 1
Variations of continuous-culture chemostat systems and flowcells for the study of mixed bacterial cultures.
To define how communities develop and change as a result of environmental insults, it was important to establish methods for achieving reproducible, complex communities of oral bacteria in the chemostat. Marsh et al. (35) did this through a series of studies involving defined inocula of nine commonly isolated organisms from supragingival plaque. Again, the details of these studies can be found elsewhere (35). Note, one can study oral microbial development using continuous-culture methods, and complex communities of organisms can be reproducibly obtained for long periods of study. These groundbreaking studies have opened the door for additional work focusing on not only the effects of environmental changes, but also on the effect of antimicrobials.
Having established the ability to study mixed populations of oral bacteria, investigators focused on the different groups of organisms associated with different oral diseases. In the field of cariology, it was of great interest to determine how communities changed in response to excess sugar and concomitant production of lactic acid by the mutans group streptococci. Studies by Bradshaw et al. (9) focused on the effects of carbohydrate pulses and pH on population shifts. Later studies looked at the effect of low fluoride concentrations on these communities of organisms (10). Collectively, these studies had a substantial impact on the field and once again demonstrated the power of being able to study microbial community development under continuous-culture conditions, i.e., controlling nutrient concentrations and growth rates.
Several studies have been conducted to look at the effects of antimicrobials on communities of oral bacteria (59). Studies by Bradshaw et al. (8) focused on the compounds triclosan and zinc citrate, alone and in combination. A novel approach was taken in these studies. The compounds were added to the chemostat by pulsing (a high initial concentration, which decreases over time) or dosing (concentration of inhibitor increases over time). This dual approach simulates how oral bacterial communities are exposed to antimicrobials in the oral cavity. Similar studies have been done with the effects of antibiotics on communities of organisms associated with periodontal disease (22). These studies found that, under certain, defined conditions, antimicrobials that had been previously characterized as broad spectrum in activity exerted more selectivity in their impact on complex communities of organisms. This increased selectivity was discovered only because of the power of the mixed, continuous-culture methodology that was used in the investigations.
The next evolution of the continuous-culture methodology to study communities of organisms has been the focus on biofilms. An enormous volume of literature still exists on the development of bacterial biofilms and their study during the past 5–10 years. Two recent volumes of Methods in Enzymology have been dedicated solely to microbial growth in biofilms and the techniques for studying them (17, 18). The importance of studying biofilms cannot be overstressed; essentially all oral diseases caused by microorganisms are a result of perturbations of the homeostasis of mixed microbial communities growing in a biofilm state (7, 32).
A classical system used to study biofilms in conjunction with continuous-culture systems is the Robbins device (25). This device is an artificial, multiport sampling catheter. It contains sampling ports whereby silicon disks are inserted in such a manner that they lie on the inner surface and are subject to the flow of media and bacteria. In many cases, this sampling system is attached to the effluent line of a chemostat so that cells growing at defined growth rates and nutrient conditions are then used as inocula for the biofilms. The biofilms can be studied through time, be exposed to antimicrobials, assayed for elaboration of various enzymes and by-products, and studied as to the response of immune cells. Thus, one has the benefit of cells grown under conditions more closely resembling those in the oral cavity and other sites in the body, and the ability to study biofilms developing under these precisely regulated conditions.
Other modified continuous-culture systems for the study of biofilms have been developed (Table 1). Bowden (1) describes a modified chemostat that has ten sampling rods that can be inserted into the lid, allowing for surfaces within the chemostat on which biofilms can form. Thus, once again, the model allows for the important environmental control of nutrient concentrations, pH, gaseous environment, and shear forces. Advantages of this model include the ability to compare equivalent biomasses of planktonic and biofilm cells grown under identical environmental conditions. One can reproducibly obtain biofilms that are equivalent from experiment to experiment.
Several other modifications of the continuous-culture system for studying biofilms have been reviewed (17, 18). Constant-depth film fermentors are similar in design to the modified chemostat described above, but with notable differences in terms of the volume of media, the sampling, and the control of the depth of biofilms (58). These systems have been used to study the effect of antimicrobials on biofilm formation, the effect of repeated pulsing of antimicrobials on biofilms, and the effect of various nutrients on the susceptibility of biofilms to antimicrobials (58).
Considerable valuable information has been gained throughout the years on the development of oral microbial communities and their response to antimicrobials. Look at the excellent, recent review by Bradshaw and Marsh (5) of the evolution of these model systems from the monoculture chemostat to the complex, mixed continuous-culture system studying gene expression in biofilms. These model systems have allowed us to determine how bacteria interact in a developing community and react to environmental stimuli. Such studies have shown how the concept of community homeostasis is critically important in the maintenance of oral health.
The chemostat has been of particular value in studies of the human oral microbiota and of other microbiota in humans (5). It provides control of environmental conditions and allows variation of one parameter at a time so that cause-and-effect relationships can be established (5), in particular, in the ecology of dental plaque in health and disease. Under highly controlled and reproducible conditions, they permit modeling of specific events that occur in vivo (5). However, they do not attempt to reproduce, or as a true model, cannot reproduce all the physical properties of the habitat (5).
Flowcells
Planktonic (free-swimming) bacteria attach, colonize, and persist on surfaces in natural, clinical, and industrial settings (2, 11, 14, 30). Once attached, they form biofilms consisting of pillarlike multicellular structures, in a hydrated polymeric matrix (13), interspersed with fluid-filled channels (14, 30). Gene expression in biofilm cells is similar to that in free-living cells, and only about 1% of genes show differential expression between these two modes of growth; about 0.5% of genes are activated and about 0.5% are repressed in biofilms (56). However, few significant differences exist (56). For example, pili and flagella are reported to be involved in attachment and microcolony formation of planktonic Pseudomonas aeruginosa, and these genes are repressed in biofilm cells (56). Many microbial communities are often composed of multiple species that interact with each other and their environment. The determination of biofilm architecture, particularly the spatial arrangement of microcolonies (clusters of cells) relative to one another, has profound implications for the function of these complex communities (14). Biofilms are implicated in the pathogenesis of cystic fibrosis pulmonary infections (53), corneal infections (50), burn wound infections (50), otitis media, periodontal disease (12, 28, 46), and implantable medical devices (12, 50).
Flowcells are currently used to cultivate and study biofilms. Expression of extracellular quorum-sensing signals (extracellular chemical signals that cue cell-density-dependent gene expression) to coordinate biofilm formation (43, 53, 55), receptors (15), metabolic interactions (48), metabolic interdependence (48), horizontal gene transfer among species (20), phylogenetic groupings, mechanisms of antimicrobial resistance (31, 56), and competition among members of the biofilm (48) can all be assessed.
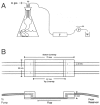
Flowcells are essentially perfusion chambers used for the observation of growth of stationary cells (Fig. 2). Two recent designs have been extensively described by Palmer and colleagues (23, 29, 47, 48). One cell is designed to observe microbial growth from the top (47). This cell has 4-mm2 parallel grooves milled into a Plexiglas base stopping a few millimeters short of the cell ends. Microscopy coverslips are then used to cover the grooves forming a closed channel. Inlet and outlet openings are bored into ends of the cell through to the channel, and tubing is then cemented into the holes. Another cell is designed to view microbial growth from either the top or bottom (47). Microscopy coverslips are used to cover both the top and the bottom of a closed channel that is formed by a molded silicon rubber gasket. Elbow-shaped inlet and outlet ports are molded out of silicon. This design has two channels and has the same dimensions as a standard microscope slide, thereby making it compatible with most microscope stage hardware.
The flowcell is connected to a medium reservoir (Fig. 2A) containing broth or other biological fluid such as saliva (47, 48). A pump, placed on the outflow tube of the flowcell, is used to draw media through the cell and into a waste collection vessel. This basic setup optimizes the results and eliminates complications such as the occurrence of air bubbles in the flowcell (frequently generated on the downstream side of the pump) and flowcell contamination. The microorganism(s) to be grown can be aseptically injected directly into the flowcell through the silicon tubing.
As biofilms form on the glass of the flowcell (Fig. 2B), they are frequently examined by various forms of microscopy. Frequently, confocal microscopy is used (47) to examine biofilms in flowcells with a Plexiglas base because transmitted light is not required. However, if cell monolayers are examined with high-resolution, transmitted-light, phase-contrast optics, then the working distance of the lens and the travel of the substage condenser are important in setting the total thickness of the flowcell, and the flowcell must have glass as the base and as the top (47). Observation of colonization on the opposite coverslip is impossible with high-magnification, oil-immersion optics because the working distances of such lenses are too small. Epifluorescence microscopy techniques are also used to observe cells and eliminate the restrictions imposed by phase-contrast Koehjler illumination (47). Each of these techniques has advantages and disadvantages that involve the size and total thickness of the flowcell; the substage condenser; and the working distance, numerical aperture, and magnification of the microscope objective lens.
Overall, flowcells offer many advantages (47). They can be made inexpensively from a variety of materials. The laminar flow of media through the cell can be controlled and induces a defined, constant environment. Finally, the flowcell construction allows for visual assessment of biofilm formation by time-resolved, nondestructive means. Affluent and effluent media can be sampled. Unfortunately, flowcells do have some disadvantages (47). Sterilization can be difficult and dependent on the common materials used for flowcell construction. Autoclaving, gas sterilization, and disinfection procedures can be used to varying levels.
Conclusion
This chapter briefly describes two methods used to investigate microbial interactions in vitro, continuous-culture chemostat systems and flowcells. Biofilms form in each of the two models that can be used to identify mechanisms of interspecies and intergeneric cooperation among microorganisms applicable to polymicrobial diseases. Chemostat studies can be used to assess the responses of steady-state cultures to stresses applied by the experimenter. The steady state can be easily perturbed and allows unequivocal data on the effect of any perturbation. Flowcells offer additional advantages and the effects of treatment on biofilms can be visually assessed by time-resolved, nondestructive means or measured in sampled, effluent waste media. Both of these systems have identified synergistic and mutualistic interactions among microorganisms resulting in unique mechanisms of attachment and metabolic interdependence. These methods are rapidly gaining acceptance and are used to study biofilm diseases, particularly the interactions among members of the resident flora, factors involved in the transition of the biofilm from a commensal to a pathogenic relationship with the host, and the mode of action of antimicrobials.
References
- 1.
- Bowden G. H. Controlled environment model for accumulation of biofilms of oral bacteria. Methods Enzymol. 1999;310:216–224. [PubMed: 10547795]
- 2.
- Bower C. K., Daeschel M. A., McGuire J. Protein antimicrobial barriers to bacterial adhesion. J. Dairy Sci. 1998;81:2771–2778. [PubMed: 9812283]
- 3.
- Bradshaw D. J., Marsh P. D. Analysis of pH-driven disruption of oral microbial communities in vitro. Caries Res. 1998;32:456–462. [PubMed: 9745120]
- 4.
- Bradshaw D. J., Marsh P. D. Effect of sugar alcohols on the composition and metabolism of a mixed culture of oral bacteria grown in a chemostat. Caries Res. 1994;28:251–256. [PubMed: 8069881]
- 5.
- Bradshaw D. J., Marsh P. D. Use of continuous flow techniques in modeling dental plaque biofilms. Methods Enzymol. 1999;310:279–296. [PubMed: 10547800]
- 6.
- Bradshaw D. J., Marsh P. D., Allison C., Schilling K. M. Effect of oxygen, inoculum composition and flow rate on development of mixed-culture oral biofilms. Microbiology. 1996;142:623–629. [PubMed: 8868437]
- 7.
- Bradshaw D. J., Marsh P. D., Schilling K. M., Cummins D. A modified chemostat system to study the ecology of oral biofilms. J. Appl. Bacteriol. 1996;80:124–130. [PubMed: 8642010]
- 8.
- Bradshaw D. J., Marsh P. D., Watson G. K., Cummins D. The effects of triclosan and zinc citrate, alone and in combination, on a community of oral bacteria grown in vitro. J. Dent. Res. 1993;72:25–30. [PubMed: 8380287]
- 9.
- Bradshaw D. J., McKee A. S., Marsh P. D. Effects of carbohydrate pulses and pH on population shifts within oral microbial communities in vitro. J. Dent. Res. 1989;68:1298–1302. [PubMed: 2674233]
- 10.
- Bradshaw D. J., McKee A. S., Marsh P. D. Prevention of population shifts in oral microbial communities in vitro by low fluoride concentrations. J. Dent. Res. 1990;69:436–441. [PubMed: 2307745]
- 11.
- Brown M. R., Barker J. Unexplored reservoirs of pathogenic bacteria: protozoa and biofilms. Trends Microbiol. 1999;7:46–50. [PubMed: 10068997]
- 12.
- Chen C. Periodontitis as a biofilm infection. J. Calif. Dent. Assoc. 2001;29:362–369. [PubMed: 11400542]
- 13.
- Costerton J. W., Stewart P. S., Greenberg E. P. Bacterial biofilms: a common cause of persistent infections. Science. 1999;284:1318–1322. [PubMed: 10334980]
- 14.
- Davey M. E., O'Toole G. A. Microbial biofilms: from ecology to molecular genetics. Microbiol. Mol. Biol. Rev. 2000;64:847–867. [PMC free article: PMC99016] [PubMed: 11104821]
- 15.
- Demuth D. R., Irvine D. C., Costerton J. W., Cook G. S., Lamont R. J. Discrete protein determinant directs the species-specific adherence of Porphyromonas gingivalis to oral streptococci. Infect. Immun. 2001;69:5736–5741. [PMC free article: PMC98690] [PubMed: 11500450]
- 16.
- Dibdin G., Wimpenny J. Steady-state biofilm: practical and theoretical models. Methods Enzymol. 1999;310:296–322. [PubMed: 10547801]
- 17.
- Doyle, R. J. (ed.). 2001. Methods in Enzymology, vol. 310. Biofilms. Academic Press, San Diego, Calif.
- 18.
- Doyle, R. J. (ed.). 2001. Methods in Enzymology, vol. 337. Microbial Growth in Biofilms. Part B: Special Environments and Physicochemical Aspects, Academic Press, San Diego, Calif.
- 19.
- Gerritse J., Schut F., Gottschal J. C. Modelling of mixed chemostat cultures of an aerobic bacterium, Comamonas testosteroni, and an anaerobic bacterium, Veillonella alcalescens: comparison with experimental data. Appl. Environ. Microbiol. 1992;58:1466–1476. [PMC free article: PMC195627] [PubMed: 1622213]
- 20.
- Ghigo J. M. Natural conjugative plasmids induce bacterial biofilm development. Nature. 2001;412:442–445. [PubMed: 11473319]
- 21.
- Herbert, D. 1961. Continuous culture of microorganisms. SCI Monograph 12. Butterworths, London, United Kingdom.
- 22.
- Hoeven J. S., van den Kiebom C. W. A. Effects of propicillin on mixed continuous cultures of periodontal bacteria. Antimicrob. Agents Chemother. 1991;35:1717–1720. [PMC free article: PMC245256] [PubMed: 1952836]
- 23.
- Jubb, K. V. F., P. C. Kennedy, and N. Palmer 1985. The respiratory system, p. 520–528. In K. V. F. Jubb, P. C. Kennedy, and N. Palmer (ed.), Pathology of Domestic Animals, 3rd ed., vol. 2. Academic Press, San Diego, Calif.
- 24.
- Keevil C. W. Continuous culture models to study pathogens in biofilms. Methods Enzymol. 2001;337:104–122. [PubMed: 11398424]
- 25.
- Kharazmi A., Giwercman B., Hoiby N. Robbins device in biofilm research. Methods Enzymol. 1999;310:207–215. [PubMed: 10547794]
- 26.
- Kinniment S. L., Wimpenny J. W., Adams D., Marsh P. D. Development of a steady-state oral microbial biofilm community using the constant-depth film fermenter. Microbiology. 1996;142:631–638. [PubMed: 8868438]
- 27.
- Kinniment S. L., Wimpenny J. W., Adams D., Marsh P. D. The effect of chlorhexidine on defined, mixed culture oral biofilms grown in a novel model system. J. Appl. Bacteriol. 1996;81:120–125. [PubMed: 8760321]
- 28.
- Kolenbrander P. E. Oral microbial communities: biofilms, interactions, and genetic systems. Annu. Rev. Microbiol. 2000;54:413–437. [PubMed: 11018133]
- 29.
- Kolenbrander P. E., Andersen R. N., Kazmerzak K., Wu R., Palmer, Jr R. J. Spatial organization of oral bacteria in biofilms. Methods Enzymol. 1999;310:322–332. [PubMed: 10547802]
- 30.
- Kuchma S. L., O'Toole G. A. Surface-induced and biofilm-induced changes in gene expression. Curr. Opin. Biotechnol. 2000;11:429–433. [PubMed: 11024358]
- 31.
- Mah T. F., O'Toole G. A. Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol. 2001;9:34–39. [PubMed: 11166241]
- 32.
- Marsh P. D. The role of microbiology in models of dental caries. Adv. Dent. Res. 1995;9:244–254. [PubMed: 8615947]
- 33.
- Marsh P. D. Sugar, fluoride, pH and microbial homeostasis in dental plaque. Proc. Finn. Dent. Soc. 1991;87:515–525. [PubMed: 1775479]
- 34.
- Marsh P. D., Bradshaw D. J. The effect of fluoride on the stability of oral bacterial communities in vitro. J. Dent. Res. 1990;69:668–671. [PubMed: 2179328]
- 35.
- Marsh P. D., Hunter J. R., Bowden G. H., Hamilton I. R., McKee A. S., Hardie J. M., Ellwood D. C. The influence of growth rate and nutrient limitation on the microbial composition and biochemical properties of a mixed culture of oral bacteria grown in a chemostat. J. Gen. Microbiol. 1983;129:755–770. [PubMed: 6348208]
- 36.
- Marsh P. D., McDermid A. S., Keevil C. W., Ellwood D. C. Effect of environmental conditions on the fluoride sensitivity of acid production by S. sanguis NCTC 7865. J. Dent. Res. 1985;64:85–89. [PubMed: 3855887]
- 37.
- Marsh P. D., McDermid A. S., Keevil C. W., Ellwood D. C. Environmental regulation of carbohydrate metabolism by Streptococcus sanguis NCTC 7865 grown in a chemostat. J. Gen. Microbiol. 1985;131:2505–2514. [PubMed: 2999295]
- 38.
- Marsh P. D., McDermid A. S., McKee A. S., Baskerville A. The effect of growth rate and haemin on the virulence and proteolytic activity of Porphyromonas gingivalis W50. Microbiology. 1994;140:861–865. [PubMed: 8012602]
- 39.
- McDermid A. S., Marsh P. D., Keevil C. W., Ellwood D. C. Additive inhibitory effects of combinations of fluoride and chlorhexidine on acid production by Streptococcus mutans and Streptococcus sanguis. Caries Res. 1985;19:64–71. [PubMed: 3856486]
- 40.
- McDermid A. S., McKee A. S., Ellwood D. C., Marsh P. D. The effect of lowering the pH on the composition and metabolism of a community of nine oral bacteria grown in a chemostat. J. Gen. Microbiol. 1986;132:1205–1214. [PubMed: 3095488]
- 41.
- McDermid A. S., McKee A. S., Marsh P. D. A mixed-culture chemostat system to predict the effect of anti-microbial agents on the oral flora: preliminary studies using chlorhexidine. J. Dent. Res. 1987;66:1315–1320. [PubMed: 3476598]
- 42.
- McKee A. S., McDermid A. S., Ellwood D. C., Marsh P. D. The establishment of reproducible, complex communities of oral bacteria in the chemostat using defined inocula. J. Appl. Bacteriol. 1985;59:263–275. [PubMed: 3932293]
- 43.
- Miller M. B., Bassler B. L. Quorum sensing in bacteria. Annu. Rev. Microbiol. 2001;55:165–199. [PubMed: 11544353]
- 44.
- Monod J. La technique de culture continue. Théorie et applications. Ann. Inst. Pasteur (Paris). 1950;79:390–410.
- 45.
- Novick A., Szilard L. Experiments with the chemostat on spontaneous mutations of bacteria. Proc. Natl. Acad. Sci. USA. 1950;36:708–719. [PMC free article: PMC1063276] [PubMed: 14808160]
- 46.
- O'Toole G., Kaplan H. B., Kolter R. Biofilm formation as microbial development. Annu. Rev. Microbiol. 2000;54:49–79. [PubMed: 11018124]
- 47.
- Palmer R. J. Jr. Microscopy flowcells: perfusion chambers for real-time study of biofilms. Methods Enzymol. 1999;310:160–166. [PubMed: 10547790]
- 48.
- Palmer R. J. Jr.,, Kazmerzak K., Hansen M. C., Kolenbrander P. E. Mutualism versus independence: strategies of mixed-species oral biofilms in vitro using saliva as the sole nutrient source. Infect. Immun. 2001;69:5794– 5804. [PMC free article: PMC98697] [PubMed: 11500457]
- 49.
- Pelczar, M. J., Jr., E. C. S. Chan, and N. R. Krieg. 1986. Microbiology, 5th ed. McGraw-Hill Publishing Company, New York, N.Y.
- 50.
- Rumbaugh K. P., Griswold J. A., Hamood A. N. The role of quorum sensing in the in vivo virulence of Pseudomonas aeruginosa. Microbes Infect. 2000;2:1721–1731. [PubMed: 11137045]
- 51.
- Saunders K. A., Greenman J. The formation of mixed culture biofilms of oral species along a gradient of shear stress. J. Appl. Microbiol. 2000;89:564–572. [PubMed: 11054158]
- 52.
- Saunders K. A., Greenman J., McKenzie C. Ecological effects of triclosan and triclosan monophosphate on defined mixed cultures of oral species grown in continuous culture. J. Antimicrob. Chemother. 2000;45:447–452. [PubMed: 10747820]
- 53.
- Singh P. K., Schaefer A. L., Parsek M. R., Moninger T. O., Welsh M. J., Greenberg E. P. Quorum-sensing signals indicate that cystic fibrosis lungs are infected with bacterial biofilms. Nature. 2000;407:762–764. [PubMed: 11048725]
- 54.
- Smalley J. W., Birss A. J., McKee A. S., Marsh P. D. Hemin regulation of hemoglobin binding by Porphyromonas gingivalis. Curr. Microbiol. 1998;36:102–106. [PubMed: 9425248]
- 55.
- Swift S., Downie J. A., Whitehead N. A., Barnard A. M., Salmond G. P., Williams P. Quorum sensing as a population-density-dependent determinant of bacterial physiology. Adv. Microb. Physiol. 2001;45:199–270. [PubMed: 11450110]
- 56.
- Whiteley M., Bangera M. G., Bumgarner R. E., Parsek M. R., Teitzel G. M., Lory S., Greenberg E. P. Gene expression in Pseudomonas aeruginosa biofilms. Nature. 2001;413:860–864. [PubMed: 11677611]
- 57.
- Whiteley M., Ott J. R., Weaver E. A., McLean R. J. Effects of community composition and growth rate on aquifer biofilm bacteria and their susceptibility to betadine disinfection. Environ. Microbiol. 2001;3:43–52. [PubMed: 11225722]
- 58.
- Wilson M. Use of constant depth film fermentor in studies of biofilms of oral bacteria. Methods Enzymol. 1999;310:264–279. [PubMed: 10547799]
- 59.
- Zelver N., Hamilton M., Pitts B., Goeres D., Walker D., Sturman P., Heersink J. Measuring antimicrobial effects on biofilm bacteria: from laboratory to field. Methods Enzymol. 1999;310:608–628. [PubMed: 10547823]
Publication Details
Author Information and Affiliations
Authors
David R. Drake1 and Kim A. Brogden2.Affiliations
Copyright
Publisher
ASM Press, Washington (DC)
NLM Citation
Drake DR, Brogden KA. Continuous-Culture Chemostat Systems and Flowcells as Methods to Investigate Microbial Interactions. In: Brogden KA, Guthmiller JM, editors. Polymicrobial Diseases. Washington (DC): ASM Press; 2002. Chapter 2.