NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004-2013.
| Chemical name: | 4-[11C]Methylphenyl-1,4-diazabicyclo[3.2.2]nonane-4-carboxylate |

|
| Abbreviated name: | [11C]CHIBA-1001 | |
| Synonym: | ||
| Agent category: | Compound | |
| Target: | Neuronal α7 nicotinic acetylcholine receptor (nAChR) | |
| Target category: | Receptor | |
| Method of detection: | PET | |
| Source of signal: | 11C | |
| Activation: | No | |
| Studies: |
| Click on the above structure for additional information in PubChem. |
Background
[PubMed]
Neuronal α4β2 nicotinic cholinergic receptors (nAChRs) are part of a heterogeneous family of ligand-gated ion channels expressed in the central nervous system, where their activation by acetylcholine and nicotine causes a rapid increase in cellular permeability to ions, such as Na+ and Ca2+ (1-3). Nicotinic receptors exist as pentamers (homomeric or heteromeric) in various brain regions and ganglia. There are nine subtypes of ligand-binding α receptors (α2 to α10) and four subtypes of structural β receptors (β2 to β5). nAChRs have been found to be involved in cognitive processes such as learning memory and control of movement in normal subjects. nAChR dysfunction has been implicated in a number of human diseases such as schizophrenia, Huntington's disease, Alzheimer's disease, and Parkinson's disease. nAChRs also play a significant role in nicotine addiction and other health problems associated with tobacco smoking.
3-[2(S)-2-Azetidinylmethoxy]pyridine (A-85380) is a highly potent and selective α4β2 nAChR agonist with subnanomolar affinity (4, 5). 6-[18F]Fluoro-A-85380 and 2-[18F]fluoro-A-85380 have been studied in humans as positron emission tomography (PET) agents for α4β2 nAChR imaging in the brain. A-85380 has also been labeled as 5-[123I]iodo-A-85380, which has been developed as a single-photon emission computed tomography agent for the non-invasive study of α4β2 nAChR in the brain. There are some implications that homomeric α7 nAChRs may play a role in the pathophysiology of neuropsychiatric disorders (6-8). α7 nAChRs are highly expressed in the cerebral cortex, hippocampus and subcortical limbic regions, which are involved in learning, memory, and information processing. 4-Bromophenyl-1,4-diazabicyclo[3.2.2]nonane-4-carboxylate (SSR180711) has been shown to be a potent and selective partial agonist for α7 nAChRs with nanomolar affinity (9). 4-[11C]Methylphenyl-1,4-diazabicyclo[3.2.2]nonane-4-carboxylate ([11C]CHIBA-1001), a 4-methyl substituted derivative of SSR180711, has been developed as a PET agent for the non-invasive study of α7 nAChR in the brain (10).
Related Resource Links:
Synthesis
[PubMed]
Hashimoto et al. (10) reported the synthesis of [11C]CHIBA-1001 by methylation of 4-(tributylstannyl)phenyl-1,4-diazabicyclo[3.2.2]nonane-4-carboxylate in the presence of CuCl2, potassium carbonate, and [11C]methyl iodide in DMF solution. The mixture was heated for 5 min at 70°C, and [11C]CHIBA-1001 was purified with high-performance liquid chromatography. Average radiochemical yield was 9.49 ± 1.45%. The radiochemical purity was >98% with a specific activity of 344 ± 36 GBq/µmol (9.3 ± 1.0 Ci/µmol; n = 12) at the end of synthesis.
In Vitro Studies: Testing in Cells and Tissues
[PubMed]
Hashimoto et al. (10) reported that CHIBA-1001 and SSR180711 exhibited IC50 values (obtained with the use of [125I]α-bungarotoxin) of 24.9 and 45.8 nM in rat brain membranes, respectively. CHIBA-1001 exhibited little affinity for α4β2, α2β4, and α3β4 nAChRs as well as other receptors.
Animal Studies
Rodents
[PubMed]
Toyohara et al. (11) performed ex vivo biodistribution of [11C]CHIBA-1001 in normal mice (n = 4/group) at 1 – 90 min. The highest uptake (percentage of injected dose per gram of tissue: %ID/g) of [11C]CHIBA-1001 was found in the lungs (23.77) followed by the kidneys (13.17), liver (17.32), pancreas (9.15), spleen (5.44), small intestines (4.23), heart (6.16), brain (5.83) and muscle (2.46) at 1 -30 min after injection. The radioactivity level of [11C]CHIBA-1001 was low in the blood (0.65) at 5 min after injection. HPLC analysis of the plasma showed that 43.6% and 21.3% of [11C] CHIBA-1001 were intact at 15 and 30 min after injection with two major hydrophilic metabolites. On the other hand, little degradation of [11C] CHIBA-1001 was detected in the brain (>99% intact) at these time points. The effective dose estimated for human adults was 3.8 µSv/MBq. No blocking studies were reported.
Non-Human Primates
[PubMed]
[11C]CHIBA-1001 PET studies with monkeys (n = 3) showed substantial brain accumulation with selective uptake in the regions of the hippocampus, cortex and basal ganglia (0.035-0.041%dose/cc)10–30 min after injection with gradual washouts (10); the cerebellum (0.022%dose/cc) showed lower binding than the other brain regions. [11C]CHIBA-1001 radioactivity in the brain regions was reduced to <0.028%dose/cc by pretreatment with the α7 nAChR agonists SSR180711 and A844606 (5.0 mg/kg, 30 min). However, the accumulation of radioactivity in the brain regions after administration of [11C]CHIBA-1001 was not affected by pretreatment with the selective α4β2 nAChR agonist A-85380 (1.0 mg/kg, 30 min). The fraction of unchanged [11C]CHIBA-1001 in the plasma as determined with high-performance liquid chromatography was ~50% at 60 min after injection.
Human Studies
[PubMed]
[11C]CHIBA-1001 (680 MBq/18.4 mCi, 17 nmol) PET scans with one healthy male subject (48 years old) showed substantial brain accumulation with selective uptake in the regions of the hippocampus, frontal cortex, thalamus, and cerebellum at 10–30 min after injection with gradual washouts (11). In Logan graphical analysis (total distribution volume, VT), the cerebellum (16.8 ml/g) showed lower binding than the thalamus (21.6 ml/g), amygdale (20.3), putamen (20.0 ml/g), hippocampus (19.7 ml/g), and frontal cortex (18.1 ml/g). The fraction of unchanged [11C]A-582941 in the plasma as determined with high-performance liquid chromatography was 92.2% at 30 min and 81.4% at 60 min after injection.
References
- 1.
- Alkondon M., Albuquerque E.X. The nicotinic acetylcholine receptor subtypes and their function in the hippocampus and cerebral cortex. Prog Brain Res. 2004;145:109–20. [PubMed: 14650910]
- 2.
- Gotti C., Riganti L., Vailati S., Clementi F. Brain neuronal nicotinic receptors as new targets for drug discovery. Curr Pharm Des. 2006;12(4):407–28. [PubMed: 16472136]
- 3.
- Hogg R.C., Raggenbass M., Bertrand D. Nicotinic acetylcholine receptors: from structure to brain function. Rev Physiol Biochem Pharmacol. 2003;147:1–46. [PubMed: 12783266]
- 4.
- Abreo M.A., Lin N.H., Garvey D.S., Gunn D.E., Hettinger A.M., Wasicak J.T., Pavlik P.A., Martin Y.C., Donnelly-roberts D.L., Anderson D.J., Sullivan J.P., Williams M., Arneric S.P., Holladay M.W. Novel 3-Pyridyl ethers with subnanomolar affinity for central neuronal nicotinic acetylcholine receptors. J Med Chem. 1996;39(4):817–25. [PubMed: 8632405]
- 5.
- Sullivan J.P., Donnelly-Roberts D., Briggs C.A., Anderson D.J., Gopalakrishnan M., Piattoni-Kaplan M., Campbell J.E., McKenna D.G., Molinari E., Hettinger A.M., Garvey D.S., Wasicak J.T., Holladay M.W., Williams M., Arneric S.P. A-85380 [3-(2(S)-azetidinylmethoxy) pyridine]: in vitro pharmacological properties of a novel, high affinity alpha 4 beta 2 nicotinic acetylcholine receptor ligand. Neuropharmacology. 1996;35(6):725–34. [PubMed: 8887981]
- 6.
- Gotti C., Zoli M., Clementi F. Brain nicotinic acetylcholine receptors: native subtypes and their relevance. Trends Pharmacol Sci. 2006;27(9):482–91. [PubMed: 16876883]
- 7.
- Nomikos G.G., Schilstrom B., Hildebrand B.E., Panagis G., Grenhoff J., Svensson T.H. Role of alpha7 nicotinic receptors in nicotine dependence and implications for psychiatric illness. Behav Brain Res. 2000;113(1-2):97–103. [PubMed: 10942036]
- 8.
- Nordberg A. Nicotinic receptor abnormalities of Alzheimer's disease: therapeutic implications. Biol Psychiatry. 2001;49(3):200–10. [PubMed: 11230871]
- 9.
- Biton B., Bergis O.E., Galli F., Nedelec A., Lochead A.W., Jegham S., Godet D., Lanneau C., Santamaria R., Chesney F., Leonardon J., Granger P., Debono M.W., Bohme G.A., Sgard F., Besnard F., Graham D., Coste A., Oblin A., Curet O., Vige X., Voltz C., Rouquier L., Souilhac J., Santucci V., Gueudet C., Francon D., Steinberg R., Griebel G., Oury-Donat F., George P., Avenet P., Scatton B. SSR180711, a novel selective alpha7 nicotinic receptor partial agonist: (1) binding and functional profile. Neuropsychopharmacology. 2007;32(1):1–16. [PubMed: 17019409]
- 10.
- Hashimoto K., Nishiyama S., Ohba H., Matsuo M., Kobashi T., Takahagi M., Iyo M., Kitashoji T., Tsukada H. [11C]CHIBA-1001 as a novel PET ligand for alpha7 nicotinic receptors in the brain: a PET study in conscious monkeys. PLoS ONE. 2008;3(9):e3231. [PMC free article: PMC2529405] [PubMed: 18800169]
- 11.
- Toyohara J., Sakata M., Wu J., Ishikawa M., Oda K., Ishii K., Iyo M., Hashimoto K., Ishiwata K. Preclinical and the first clinical studies on [11C]CHIBA-1001 for mapping alpha7 nicotinic receptors by positron emission tomography. Ann Nucl Med. 2009;23(3):301–9. [PubMed: 19337782]
- PMCPubMed Central citations
- PubChem SubstanceRelated PubChem Substances
- PubMedLinks to PubMed
- Review 4-[5-(4-[(18)F]Fluoro-phenyl)-[1,3,4]oxadiazol-2-yl]-1,4-diaza-bicyclo[3.2.2]nonane.[Molecular Imaging and Contrast...]Review 4-[5-(4-[(18)F]Fluoro-phenyl)-[1,3,4]oxadiazol-2-yl]-1,4-diaza-bicyclo[3.2.2]nonane.Leung K. Molecular Imaging and Contrast Agent Database (MICAD). 2004
- Review 2-(5-[(11)C]Methyl-hexahydro-pyrrolo[3,4-c]pyrrol-2-yl)-xanthene-9-one.[Molecular Imaging and Contrast...]Review 2-(5-[(11)C]Methyl-hexahydro-pyrrolo[3,4-c]pyrrol-2-yl)-xanthene-9-one.Leung K. Molecular Imaging and Contrast Agent Database (MICAD). 2004
- Review 2-[(11)C]Methyl-5-[6-phenylpyridazine-3-yl]octahydropyrrolo[3,4-c]pyrrole.[Molecular Imaging and Contrast...]Review 2-[(11)C]Methyl-5-[6-phenylpyridazine-3-yl]octahydropyrrolo[3,4-c]pyrrole.Leung K. Molecular Imaging and Contrast Agent Database (MICAD). 2004
- Review 4-[(76)Br]Bromophenyl-1,4-diazabicyclo(3.2.2) nonane-4-carboxylate.[Molecular Imaging and Contrast...]Review 4-[(76)Br]Bromophenyl-1,4-diazabicyclo(3.2.2) nonane-4-carboxylate.Leung K. Molecular Imaging and Contrast Agent Database (MICAD). 2004
- Review 5-[(123)I]Iodo-3-[2(S)-2-azetidinylmethoxy]pyridine.[Molecular Imaging and Contrast...]Review 5-[(123)I]Iodo-3-[2(S)-2-azetidinylmethoxy]pyridine.Leung K. Molecular Imaging and Contrast Agent Database (MICAD). 2004
- 4-[11C]Methylphenyl-1,4-diazabicyclo[3.2.2]nonane-4-carboxylate - Molecular Imag...4-[11C]Methylphenyl-1,4-diazabicyclo[3.2.2]nonane-4-carboxylate - Molecular Imaging and Contrast Agent Database (MICAD)
Your browsing activity is empty.
Activity recording is turned off.
See more...

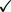 In vitro
In vitro