By agreement with the publisher, this book is accessible by the search feature, but cannot be browsed.
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Gilbert SF. Developmental Biology. 6th edition. Sunderland (MA): Sinauer Associates; 2000.

Developmental Biology. 6th edition.
Show detailsFusion of genetic material in sea urchins
In sea urchins, the sperm nucleus enters the egg perpendicular to the egg surface. After fusion of the sperm and egg plasma membranes, the sperm nucleus and its centriole separate from the mitochondria and the flagellum. The mitochondria and the flagellum disintegrate inside the egg, so very few, if any, sperm-derived mitochondria are found in developing or adult organisms (Dawid and Blackler 1972; Sutovsky et al. 1999). In mice, it is estimated that only 1 out of every 10,000 mitochondria is sperm-derived (Gyllensten et al. 1991). Thus, although each gamete contributes a haploid genome to the zygote, the mitochondrial genome is transmitted primarily by the maternal parent. Conversely, in almost all animals studied (the mouse being the major exception), the centrosome needed to produce the mitotic spindle of the subsequent divisions is derived from the sperm centriole (Sluder et al. 1989, 1993).
The egg nucleus, once it is haploid, is called the female pronucleus. Once inside the egg, the sperm nucleus decondenses to form the male pronucleus. The sperm nucleus undergoes a dramatic transformation. The nuclear envelope vesiculates into small packets, thereby exposing the compact sperm chromatin to the egg cytoplasm (Longo and Kunkle 1978; Poccia and Collas 1997). The proteins holding the sperm chromatin in its condensed, inactive state are exchanged for other proteins derived from the egg cytoplasm. This exchange permits the decondensation of the sperm chromatin. In sea urchins, decondensation appears to be initiated by the phosphorylation of two sperm-specific histones that bind tightly to the DNA. This process begins when the sperm comes into contact with a glycoprotein in the egg jelly that elevates the level of cAMP-dependent protein kinase activity. These protein kinases phosphorylate several of the basic residues of the sperm-specific histones and thereby interfere with their binding to DNA (Garbers et al. 1980, 1983; Porter and Vacquier 1986). This loosening is thought to facilitate the replacement of the sperm-specific histones with other histones that have been stored in the oocyte cytoplasm (Poccia et al. 1981; Green and Poccia 1985). Once decondensed, the DNA can begin transcription and replication.
After the sea urchin sperm enters the egg cytoplasm, the male pronucleus rotates 180° so that the sperm centriole is between the sperm pronucleus and the egg pronucleus. The sperm centriole then acts as a microtubule organizing center, extending its own microtubules and integrating them with egg microtubules to form an aster.* These microtubules extend throughout the egg and contact the female pronucleus, and the two pronuclei migrate toward each other (Hamaguchi and Hiramoto 1980; Bestor and Schatten 1981). Their fusion forms the diploid zygote nucleus (Figure 7.31). The initiation of DNA synthesis can occur either in the pronuclear stage (during migration) or after the formation of the zygote nucleus.
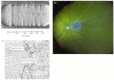
Figure 7.31
Nuclear events in the fertilization of the sea urchin. (A) Sequential photographs showing the migration of the egg pronucleus and the sperm pronucleus toward each other in an egg of Clypeaster japonicus. The sperm pronucleus is surrounded by its aster (more...)
Fusion of genetic material in mammals
In mammals, the process of pronuclear migration takes about 12 hours, compared with less than 1 hour in the sea urchin. The mammalian sperm enters almost tangentially to the surface of the egg rather than approaching it perpendicularly, and it fuses with numerous microvilli (see Figure 7.20). The mammalian sperm nucleus also breaks down as its chromatin decondenses and is then reconstructed by coalescing vesicles. The DNA of the sperm nucleus is bound by basic proteins called protamines, and these nuclear proteins are tightly compacted through disulfide bonds. In the egg cytoplasm, glutathione reduces these disulfide bonds and allows the uncoiling of the sperm chromatin (Calvin and Bedford 1971; Kvist et al. 1980; Perreault et al. 1988). The mammalian male pronucleus enlarges while the oocyte nucleus completes its second meiotic division (Figure 7.32A). The centrosome (new centriole) accompanying the male pronucleus produces its asters (largely from proteins stored in the oocyte) and contacts the female pronucleus. Then each pronucleus migrates toward the other, replicating its DNA as it travels. Upon meeting, the two nuclear envelopes break down (Figure 7.32B). However, instead of producing a common zygote nucleus (as happens in sea urchin fertilization), the chromatin condenses into chromosomes that orient themselves on a common mitotic spindle (Figure 7.32C). Thus, a true diploid nucleus in mammals is first seen not in the zygote, but at the 2-cell stage.
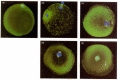
Figure 7.32
Pronuclear movements during human fertilization. The microtubules have been stained green, while the DNA is dyed blue. (A) The mature unfertilized oocyte completes the first meiotic division, budding off a polar body. (B) As the sperm enters the oocyte (more...)
WEBSITE
7.10 Sperm decondensation. The formation of the male pronucleus involves changes in both the chromatin and the nuclear envelope. The nuclear annulus may be critical to uncoiling the DNA. http://www.devbio.com/chap07/link0710.shtml
Box
The Nonequivalence of Mammalian Pronuclei.
Footnotes
- *
When Oscar Hertwig observed this radial array of sperm asters forming in his newly fertilized sea urchin eggs, he called it “the sun in the egg,” and he thought it the happy indication of a successful fertilization (Hertwig 1877). More recently, Simerly and co-workers (1994) found that certain types of human male infertility are due to defects in the centriole’s ability to form these microtubular asters. This deficiency causes the failure of pronuclear migration and the cessation of further development.
- Fusion of the Genetic Material - Developmental BiologyFusion of the Genetic Material - Developmental Biology
Your browsing activity is empty.
Activity recording is turned off.
See more...